Graph Neural Networks for Molecular Property Prediction: A Complete Guide for Drug Discovery
This article provides a comprehensive introduction to Graph Neural Networks (GNNs) for molecular property prediction, a transformative technology accelerating drug discovery and materials design.
Graph Neural Networks for Molecular Property Prediction: A Complete Guide for Drug Discovery
Abstract
This article provides a comprehensive introduction to Graph Neural Networks (GNNs) for molecular property prediction, a transformative technology accelerating drug discovery and materials design. We explore the foundational principles that make GNNs uniquely suited for modeling molecular graphs, where atoms are nodes and bonds are edges. The guide details core GNN architectures—including GCN, GAT, GIN, and emerging Kolmogorov-Arnold Networks (KANs)—and their specific applications in predicting bioactivity, toxicity, and physicochemical properties. It further addresses critical real-world challenges such as data scarcity through few-shot learning techniques and provides a framework for rigorous model validation and benchmarking against standardized datasets. Aimed at researchers, scientists, and development professionals, this resource synthesizes current methodologies, optimization strategies, and comparative analyses to empower the effective implementation of GNNs in biomedical research.
Why Graphs? The Foundational Bridge Between Molecules and Machine Learning
In the field of computer-aided drug discovery and materials science, the accurate prediction of molecular properties is a crucial task. The molecular graph paradigm, which represents atoms as nodes and bonds as edges in a graph structure, has emerged as a powerful framework for this purpose [1]. This approach provides a natural and expressive representation that allows machine learning models to directly learn from the intrinsic topological structure of molecules. Graph Neural Networks (GNNs) have particularly revolutionized this domain by enabling end-to-end learning from molecular graphs, significantly reducing reliance on manual feature engineering and opening new frontiers in molecular property prediction research [2] [3].
Molecular Representation in Graph Form
Graph Construction Fundamentals
In a molecular graph, each atom is represented as a node, characterized by features such as atomic number, chirality, formal charge, and whether it is part of a ring structure. Chemical bonds between atoms form the edges, annotated with properties including bond type (single, double, triple) and conjugation [4]. This representation preserves the fundamental connectivity and functional relationships that define a molecule's chemical identity and behavior.
The translation from chemical structure to graph typically begins with a Simplified Molecular-Input Line-Entry System (SMILES) string, which is subsequently processed using toolkits like RDKit to generate the corresponding graph object [4]. This conversion establishes a standardized pipeline for preparing molecular data for GNN models.
Advanced Representations
While the basic node-edge model captures covalent bonding relationships, recent advancements have incorporated non-covalent interactions and 3D geometric information to create more expressive representations [5] [2]. These enriched representations have demonstrated notable performance improvements, particularly for properties sensitive to spatial molecular conformation.
Table 1: Standard Molecular Graph Datasets for Benchmarking
| Dataset Name | # Graphs | Avg. Nodes/Graph | Avg. Edges/Graph | Task Type | Primary Metric |
|---|---|---|---|---|---|
| ogbg-molhiv | 41,127 | 25.5 | 27.5 | Binary classification | ROC-AUC |
| ogbg-molpcba | 437,929 | 26.0 | 28.1 | 128 binary classification tasks | Average Precision |
| QM9 | ~134,000 | ~18.0 | ~18.0 | Regression (quantum properties) | MAE |
| ClinTox | 1,478 | - | - | Binary classification | ROC-AUC |
Graph Neural Network Architectures for Molecular Property Prediction
Core Architectural Concepts
GNNs operate on molecular graphs through a message-passing framework, where nodes iteratively aggregate information from their neighbors and update their own representations [6]. This fundamental mechanism allows the network to capture both local atomic environments and global molecular structure. Several specialized architectures have been developed to optimize this process for molecular tasks:
- Graph Isomorphism Networks (GIN): Employ powerful aggregation functions to capture local substructures effectively, serving as strong baselines for 2D topological analysis [2].
- Equivariant GNNs (EGNN): Integrate 3D coordinate information while preserving Euclidean symmetries (translation, rotation, reflection), making them particularly valuable for geometry-sensitive properties [2].
- Graph Transformer Models: Incorporate global attention mechanisms to model long-range dependencies within molecules, even without explicit 3D information [2].
Emerging Hybrid Architectures
Recent research has explored hybrid models that combine the strengths of different paradigms. Kolmogorov-Arnold GNNs (KA-GNNs) integrate Fourier-based KAN modules into the three fundamental components of GNNs: node embedding, message passing, and readout [5]. This approach replaces conventional multi-layer perceptrons with learnable univariate functions based on Fourier series, enhancing both expressivity and interpretability while effectively capturing both low-frequency and high-frequency structural patterns in graphs [5].
Another innovative direction involves augmenting GNNs with knowledge from Large Language Models (LLMs), where domain-relevant knowledge and structural features are fused to create more robust molecular representations [3]. This integration helps address the long-tail distribution of molecular knowledge in LLMs by combining their conceptual understanding with structural information from GNNs.
Experimental Frameworks and Benchmarking
Standardized Evaluation Protocols
Rigorous evaluation of molecular property prediction models requires standardized benchmarks and appropriate dataset splits. The scaffold split, which groups molecules based on their two-dimensional structural frameworks, provides a more realistic assessment of model generalization compared to random splits [4]. This approach tests a model's ability to extrapolate to structurally novel compounds, mirroring real-world discovery scenarios where models must predict properties for chemically distinct molecules.
Performance metrics are tailored to task characteristics: ROC-AUC for balanced binary classification, Average Precision (AP) for highly imbalanced classification tasks, and Mean Absolute Error (MAE) for regression tasks [4].
Table 2: Performance Comparison of GNN Architectures on Molecular Property Prediction
| Model Architecture | ogbg-molhiv (ROC-AUC) | log Kow (MAE) | log Kaw (MAE) | OGB-MolHIV (ROC-AUC) |
|---|---|---|---|---|
| Graph Isomorphism Network (GIN) | 0.763 (reported in related studies) | 0.29 | 0.41 | 0.763 |
| Equivariant GNN (EGNN) | - | 0.21 | 0.25 | - |
| Graphormer | 0.807 (reported in related studies) | 0.18 | 0.28 | 0.807 |
| KA-GNN (Kolmogorov-Arnold) | Consistently outperforms conventional GNNs (exact values dataset-dependent) [5] | - | - | - |
Addressing Data Scarcity Through Multi-Task Learning
Data scarcity remains a significant challenge in molecular property prediction, particularly for specialized domains with limited experimental measurements. Multi-task Learning (MTL) has emerged as a promising strategy to leverage correlations among related molecular properties, thereby improving data efficiency [6].
However, conventional MTL approaches can suffer from negative transfer, where updates from one task detrimentally affect another. Recent work has introduced Adaptive Checkpointing with Specialization (ACS), a training scheme that mitigates this issue by combining a shared, task-agnostic backbone with task-specific heads [6]. This approach checkpoints model parameters when negative transfer signals are detected, preserving the benefits of inductive transfer while protecting individual tasks from detrimental parameter updates. The ACS method has demonstrated particular utility in ultra-low data regimes, achieving accurate predictions with as few as 29 labeled samples in sustainable aviation fuel property prediction [6].
Advanced Interpretability and Functional Group Analysis
Interpretable GNN Architectures
Model interpretability is crucial for scientific discovery and drug development, as it provides insights into the structural determinants of molecular properties. FragNet represents a significant advancement in this area, offering interpretability at four distinct levels: atoms, bonds, molecular fragments, and connections between fragments [7]. This multi-level interpretability helps researchers identify which substructures are significant for predicting specific molecular properties, facilitating scientific insight and hypothesis generation.
Similarly, KA-GNNs provide enhanced interpretability by highlighting chemically meaningful substructures through their learnable activation functions [5]. The Fourier-based KAN modules enable more transparent reasoning about which molecular patterns contribute most strongly to property predictions.
Functional Group-Centric Reasoning
Functional groups—specific groups of atoms that impart characteristic chemical properties—provide a natural bridge between molecular structure and property prediction. The recently introduced FGBench dataset enables molecular property reasoning at the functional group level, containing 625K molecular property reasoning problems with precise functional group annotations and localization [8].
This approach mirrors the reasoning process of human chemists, who typically analyze property changes through three steps: associating similar molecules, observing functional group differences, and rephrasing the problem using prior knowledge of functional groups [8]. By incorporating this fine-grained information, models can develop more interpretable, structure-aware reasoning capabilities that align with chemical intuition.
Essential Research Reagents and Computational Tools
Table 3: Essential Computational Tools for Molecular Graph Research
| Tool Name | Type | Primary Function | Application Context |
|---|---|---|---|
| RDKit | Cheminformatics Library | SMILES to graph conversion, molecular descriptor calculation | Preprocessing molecular data, feature generation [4] |
| Open Graph Benchmark (OGB) | Benchmarking Suite | Standardized datasets (e.g., ogbg-molhiv, ogbg-molpcba) and evaluation | Model benchmarking and comparison [4] |
| PyTorch Geometric | Deep Learning Library | GNN model implementation and training | Building and experimenting with GNN architectures [4] |
| DGL | Deep Learning Library | Graph neural network implementation | Scalable GNN training on large molecular datasets [4] |
| OMol25 | Quantum Chemistry Dataset | High-accuracy DFT calculations for biomolecules, metal complexes | Training and validating foundational atomistic models [9] |
| Universal Model for Atoms (UMA) | Foundational Model | Machine learning interatomic potential | Accurate prediction of atomic interactions across materials [9] |
| FGBench | Specialized Dataset | Functional group-level property reasoning | Enhancing interpretability and structure-aware reasoning [8] |
The molecular graph paradigm continues to evolve with several promising research directions. 3D-aware GNN architectures that explicitly incorporate spatial geometry are showing superior performance for physics-sensitive properties like partition coefficients [2]. The integration of external knowledge sources through LLMs and knowledge graphs addresses the long-tail challenge of molecular data while enhancing model interpretability [3]. Furthermore, foundational models pre-trained on massive diverse molecular datasets, such as Meta's Universal Model for Atoms, are demonstrating remarkable transfer learning capabilities across diverse molecular tasks [9].
In conclusion, the representation of molecules as graphs with atoms as nodes and bonds as edges has established itself as a powerful paradigm for molecular property prediction. By directly encoding molecular topology into machine learning models, this approach has enabled significant advances in accuracy, interpretability, and data efficiency. As architectural innovations continue to emerge and computational resources grow, GNNs based on this paradigm are poised to play an increasingly central role in accelerating scientific discovery and molecular design across pharmaceuticals, materials science, and environmental chemistry.
Graph Neural Networks (GNNs) have emerged as a transformative technology for molecular property prediction, enabling researchers to learn directly from graph-structured representations of chemical compounds. This technical guide provides an in-depth examination of the three core mechanics underpinning modern GNNs: message passing, aggregation, and readout. Framed within the context of drug discovery research, we detail the mathematical foundations, architectural variants, and experimental methodologies that allow GNNs to capture complex molecular patterns for accurate property prediction. By integrating recent advances such as Kolmogorov-Arnold Networks and multi-level fusion approaches, this work equips computational researchers and drug development professionals with the technical understanding necessary to leverage and advance GNN architectures in molecular machine learning.
In computational drug discovery, molecules are naturally represented as graphs where atoms serve as nodes and chemical bonds as edges. This representation makes Graph Neural Networks particularly well-suited for molecular property prediction, as they operate directly on this relational structure [10] [11]. Unlike traditional neural networks designed for grid-like or sequential data, GNNs excel at capturing the complex topological features and dependencies inherent in molecular graphs [12]. The core innovation enabling this capability is a framework known as message passing, which allows nodes to iteratively exchange information with their neighbors, effectively learning representations that encode both local atomic environments and global molecular structure [13] [14].
The significance of GNNs in molecular research is demonstrated by their widespread adoption across various pharmaceutical applications, from predicting protein-ligand binding affinities to simulating molecular interactions [5] [1]. These models have fundamentally changed molecular structural and property analysis, ushering in a new era of data-driven drug design and discovery [5]. This technical guide examines the foundational mechanics of message passing, aggregation, and readout that enable these advancements, with particular emphasis on their implementation and optimization for molecular property prediction tasks.
Foundational Concepts and Mathematical Framework
Graph Representation of Molecules
In molecular graphs, we formally define a graph as (G = (V, E)), where (V) represents the set of nodes (atoms) and (E) represents the set of edges (chemical bonds) [15]. Each node (v \in V) is associated with a feature vector (Xv) encapsulating atomic attributes such as element type, charge, and hybridization state. Similarly, edges may possess feature vectors (e{uv}) describing bond characteristics including type, length, and stereochemistry [14]. The graph structure is typically represented through an adjacency matrix (A), where (A_{ij} = 1) if nodes (i) and (j) are connected, and 0 otherwise [10].
The Message Passing Framework
The message passing framework, also referred to as Message Passing Neural Networks (MPNNs), forms the computational backbone of GNNs [14]. This iterative process enables nodes to incorporate information from their local neighborhoods, with each iteration extending the receptive field by one hop [11]. The framework consists of three fundamental operations executed sequentially at each layer:
- Message Creation: Each node computes messages to be sent to its neighbors
- Message Aggregation: Nodes collect and combine messages from neighbors
- Node Update: Nodes update their representations using aggregated messages [13]
Mathematically, for a node (i) at layer (l+1), the message passing process can be formalized as:
[ \begin{aligned} m{ij}^{(l)} &= \text{Message}(hi^{(l)}, hj^{(l)}, e{ij}) \quad \text{for } j \in N(i) \ mi^{(l)} &= \text{Aggregate}({m{ij}^{(l)} : j \in N(i)}) \ hi^{(l+1)} &= \text{Update}(hi^{(l)}, m_i^{(l)}) \end{aligned} ]
Where (hi^{(l)}) is the feature vector of node (i) at layer (l), (N(i)) is the set of neighbors of node (i), and (e{ij}) is the edge feature between nodes (i) and (j) [13].
Table 1: Components of the Message Passing Framework
| Component | Mathematical Function | Role in Molecular Context |
|---|---|---|
| Message | (m{ij}^{(l)} = M(hi^{(l)}, hj^{(l)}, e{ij})) | Encodes interaction between adjacent atoms |
| Aggregate | (mi^{(l)} = \sum{j \in N(i)} m_{ij}^{(l)}) | Combines information from bonded neighbors |
| Update | (hi^{(l+1)} = U(hi^{(l)}, m_i^{(l)})) | Updates atomic representation with local context |
The following diagram illustrates the complete message passing process between two nodes in a molecular graph:
Core Mechanic I: Message Passing in Depth
Message Functions
The message function (M(\cdot)) transforms neighbor information into a transferable format. In molecular graphs, this function encodes the relationship between adjacent atoms and their bonding characteristics [14]. The design of the message function varies across GNN architectures:
Linear Transformation: Simple yet effective, using weight matrices and biases: [ m{ij}^{(l)} = W{\text{msg}} \cdot [hj^{(l)} \| hi^{(l)} \| e{ij}] + b{\text{msg}} ] where (\|) denotes concatenation [13].
Edge-Aware Functions: Incorporate bond features directly, particularly important for distinguishing single, double, and triple bonds in molecular graphs [14].
Kolmogorov-Arnold Networks (KANs): Recent advances replace traditional linear transformations with learnable univariate functions based on the Kolmogorov-Arnold representation theorem, offering improved expressivity and parameter efficiency [5].
Neighborhood Sampling Strategies
For large molecular graphs, complete neighborhood aggregation can be computationally expensive. Several sampling strategies address this challenge:
Full Neighborhood Aggregation: Utilizes all adjacent atoms, preserving complete local chemical environment information [13].
GraphSAGE Sampling: Uniformly samples a fixed number of neighbors to maintain computational consistency [15].
Attention-Based Sampling: Dynamically selects important neighbors based on learned attention weights [10].
Core Mechanic II: Aggregation Operations
Aggregation Functions
The aggregation function combines multiple incoming messages into a single fixed-size vector. Common approaches include:
Sum Aggregation: Element-wise summation of neighbor messages, which preserves the complete neighborhood information and is permutation invariant [13] [15].
Mean Aggregation: Element-wise averaging, providing normalization for nodes with varying degrees [13].
Max Pooling: Element-wise maximum operation, capturing the most salient features from neighbors [13] [15].
Attention-Based Aggregation: Weighted combination where importance weights are learned dynamically, allowing the model to focus on more relevant neighbors [13] [10].
Table 2: Comparison of Aggregation Functions for Molecular Graphs
| Aggregation Type | Mathematical Form | Advantages in Molecular Context | Limitations |
|---|---|---|---|
| Sum | (mi = \sum{j \in N(i)} m_{ij}) | Preserves molecular bond count information | Sensitive to node degree |
| Mean | (mi = \frac{1}{|N(i)|} \sum{j \in N(i)} m_{ij}) | Normalizes for atom connectivity | May dilute strong signals |
| Max | (mi = \max{j \in N(i)} m_{ij}) | Identifies most influential interactions | Loses collective neighborhood information |
| Attention | (mi = \sum{j \in N(i)} \alpha{ij} m{ij}) | Adaptively weights atomic interactions | Increased computational complexity |
Advanced Aggregation Techniques
Recent research has introduced sophisticated aggregation mechanisms tailored for molecular property prediction:
Multi-Level Fusion: Integrates both local atomic environments and global molecular structures through simultaneous aggregation at multiple topological levels [16].
Fourier-Based KAN Aggregation: Employs Fourier series as basis functions within KAN modules to capture both low-frequency and high-frequency structural patterns in molecular graphs, enhancing representation of periodic molecular properties [5].
Graph Attention Networks (GAT): Implement attention mechanisms where attention weights (\alpha{ij}) are computed as: [ \alpha{ij} = \frac{\exp(\text{LeakyReLU}(a^T[Whi \| Whj]))}{\sum{k \in N(i)} \exp(\text{LeakyReLU}(a^T[Whi \| Wh_k]))} ] allowing each molecular node to attend to its neighbors with varying degrees of importance [10].
Core Mechanic III: Readout Functions
Graph-Level Readout for Molecular Property Prediction
The readout (or pooling) function generates graph-level representations from updated node embeddings, essential for molecular property prediction where the target property is a function of the entire molecular structure [15] [14]. Common readout operations include:
Sum/Mean/Max Readout: Simple permutation-invariant operations that combine node embeddings: [ hG = \sum{v \in V} hv^{(L)} \quad \text{or} \quad hG = \frac{1}{|V|} \sum{v \in V} hv^{(L)} \quad \text{or} \quad hG = \max{v \in V} h_v^{(L)} ] where (L) is the final GNN layer [15].
Hierarchical Readout: Performs pooling at multiple topological scales to capture both local functional groups and global molecular architecture [16].
Attention-Based Readout: Uses learned attention weights to emphasize chemically significant atoms in the final representation: [ hG = \sum{v \in V} \betav hv^{(L)}, \quad \betav = \frac{\exp(w^T hv^{(L)})}{\sum{u \in V} \exp(w^T hu^{(L)})} ] where (\beta_v) represents the importance of atom (v) to the molecular property [15].
Advanced Readout Architectures
For complex molecular properties, specialized readout architectures have demonstrated superior performance:
Fourier-KAN Readout: Replaces traditional MLP readout functions with Fourier-based Kolmogorov-Arnold Networks, providing stronger approximation capabilities for complex molecular property functions [5].
Interaction-Based Readout: Incorporates cross-modal interactions between different molecular representations (e.g., graph embeddings and molecular fingerprints) before final prediction [16].
Multi-Task Readout: Generates multiple property predictions simultaneously while sharing representation learning, particularly valuable in early drug discovery where multiple molecular characteristics need evaluation [1].
Experimental Protocols and Methodologies
Benchmarking GNN Architectures for Molecular Property Prediction
Rigorous experimental evaluation is essential for assessing GNN performance on molecular tasks. Standard protocols include:
Dataset Selection: Utilizing established molecular benchmarks such as MoleculeNet, which includes datasets for various properties like ESOL (solubility), FreeSolv (hydration free energy), and Tox21 (toxicity) [1].
Evaluation Metrics: Employing task-appropriate metrics including Root Mean Square Error (RMSE) for regression tasks, Area Under the ROC Curve (AUC-ROC) for classification tasks, and Mean Average Precision (MAP) for multi-label classification [5] [16].
Baseline Models: Comparing against traditional machine learning approaches (Random Forests, Support Vector Machines) and molecular descriptors (Morgan fingerprints) to quantify GNN advantages [1].
KA-GNN Experimental Framework
Recent work on Kolmogorov-Arnold GNNs (KA-GNNs) provides a state-of-the-art experimental framework:
Architecture Variants: Implementing both KA-GCN (KAN-augmented Graph Convolutional Networks) and KA-GAT (KAN-augmented Graph Attention Networks) to evaluate KAN integration across different GNN backbones [5].
Ablation Studies: Systematically removing KAN components from node embedding, message passing, and readout to isolate their individual contributions to performance [5].
Interpretability Analysis: Visualizing learned KAN basis functions to identify chemically meaningful molecular substructures and patterns that drive predictions [5].
The following diagram illustrates the architecture of a KA-GNN integrating KAN modules into all core components:
Table 3: Research Reagent Solutions for Molecular GNN Experiments
| Component | Function in Molecular GNN Research | Example Implementations |
|---|---|---|
| Deep Learning Frameworks | Provides foundational tensor operations and automatic differentiation | PyTorch, TensorFlow |
| GNN Libraries | Offers optimized implementations of GNN layers and graph operations | PyTorch Geometric, Deep Graph Library (DGL) |
| Molecular Datasets | Standardized benchmarks for evaluating molecular property prediction | MoleculeNet, ZINC, QM9 |
| Cheminformatics Tools | Processes molecular structures into graph representations | RDKit, OpenBabel |
| KAN Implementations | Provides Kolmogorov-Arnold Network layers for integration into GNNs | PyKAN, KAN-Torch |
Results and Performance Analysis
Quantitative Evaluation of GNN Components
Experimental results across multiple molecular benchmarks demonstrate the impact of different message passing, aggregation, and readout designs:
Aggregation Function Performance: Attention-based aggregation consistently outperforms simple sum/mean/max operations on molecular classification tasks, with average improvements of 3-5% in AUC-ROC scores across Tox21 and MUV datasets [10] [16].
Message Passing Depth: Optimal performance typically occurs at 3-5 message passing layers, balancing local chemical environment capture with over-smoothing effects [13] [15].
KA-GNN Advantages: Kolmogorov-Arnold GNNs demonstrate superior accuracy and computational efficiency compared to conventional GNNs, achieving 5-15% improvement on regression tasks like solubility and energy prediction while using 20-30% fewer parameters [5].
Case Study: Multi-Level Fusion GNN
The Multi-Level Fusion Graph Neural Network (MLFGNN) represents the state-of-the-art in molecular property prediction by integrating:
Local and Global Dependency Modeling: Simultaneously capturing atomic-level interactions through Graph Attention Networks and molecular-level patterns via Graph Transformers [16].
Multi-Modal Fusion: Incorporating molecular fingerprints as complementary features to graph representations, with adaptive fusion mechanisms [16].
Interpretable Predictions: Identifying chemically meaningful substructures that contribute to property predictions, validated by domain experts [5] [16].
Experimental results on seven benchmark datasets show that MLFGNN consistently outperforms baseline methods in both classification and regression tasks, with particularly strong performance on complex properties like drug efficacy and toxicity [16].
The core mechanics of message passing, aggregation, and readout form the computational foundation of modern Graph Neural Networks for molecular property prediction. Through iterative neighborhood information exchange, sophisticated aggregation schemes, and hierarchical readout functions, GNNs effectively capture the complex structural determinants of molecular properties. Recent advances such as Kolmogorov-Arnold Networks and multi-level fusion architectures further enhance the representational power, efficiency, and interpretability of these models.
Future research directions include developing more dynamic message passing schemes that adapt to molecular context, creating specialized aggregation functions for capturing non-covalent interactions, and designing hierarchical readout operations that explicitly model molecular substructures at multiple scales. As these technical innovations mature, GNNs will continue to transform computational drug discovery, enabling more accurate, efficient, and interpretable molecular property prediction.
Graph Neural Networks (GNNs) have emerged as a transformative technology for molecular property prediction, a critical task in modern drug discovery and materials science [1] [17]. Unlike traditional convolutional neural networks designed for grid-like data such as images, GNNs specialize in processing graph-structured data where entities (nodes) are connected by relationships (edges) [18]. This capability makes them uniquely suited for representing molecular structures, where atoms serve as nodes and chemical bonds as edges [19]. The inherent ability of GNNs to learn from both node features and topological relationships has positioned them as powerful tools for predicting molecular properties including solubility, toxicity, and biological activity [1] [17].
This technical guide provides an in-depth examination of four foundational GNN architectures that have proven particularly effective for molecular property prediction: Graph Convolutional Networks (GCN), Graph Attention Networks (GAT), Graph Isomorphism Networks (GIN), and Message Passing Neural Networks (MPNN). We explore their architectural principles, implementation methodologies, and comparative performance across various molecular prediction tasks, with a specific focus on their application within pharmaceutical research and development contexts.
Core Architectural Principles
The Message-Passing Framework
Most modern GNNs operate on a message-passing paradigm, where information is iteratively exchanged and aggregated between neighboring nodes in a graph [17] [18]. In this framework, each node updates its representation by combining its current state with aggregated information from its neighbors. This process enables nodes to incorporate contextual information from their local graph neighborhoods, with each iteration extending the receptive field by one hop [18]. The message-passing mechanism can be formally described through three key functions:
- Message Function: Defines what information is sent between nodes
- Aggregation Function: Specifies how incoming messages are combined
- Update Function: Determines how a node updates its state based on aggregated messages
This fundamental mechanism provides the foundation upon which the specialized architectures of GCN, GAT, GIN, and MPNN are built.
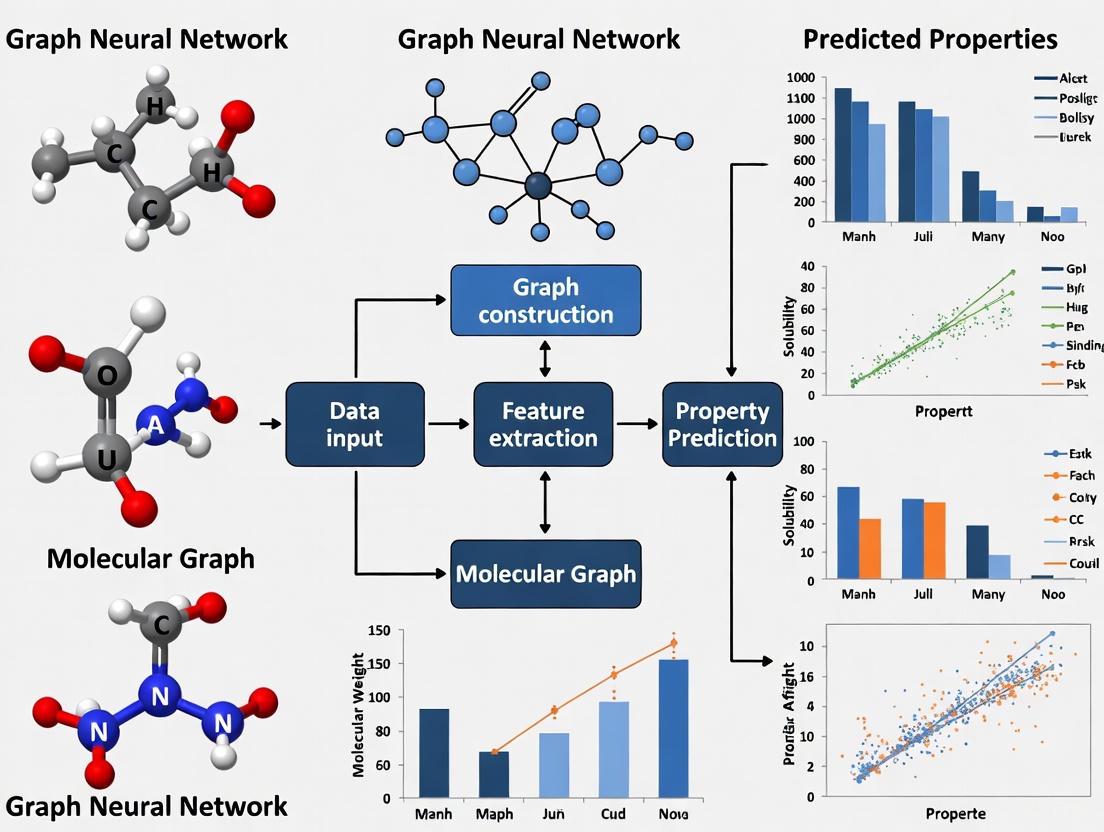
Figure 1: High-level abstraction of the GNN message-passing framework for molecular property prediction.
Molecular Graph Representation
In computational chemistry, molecules are naturally represented as graphs where atoms correspond to nodes and chemical bonds to edges [19]. Each atom node contains feature information such as atom type, hybridization state, and formal charge, while bond edges contain features such as bond type, conjugation, and stereochemistry [19] [17]. This representation allows GNNs to learn patterns directly from the structural composition of molecules, capturing complex relationships that traditional descriptor-based methods might miss [17].
Figure 2: Molecular graph representation process from chemical structure to GNN-processable format.
Architectural Blueprints
Graph Convolutional Network (GCN)
GCNs adapt convolutional operations from traditional CNNs to graph-structured data by performing localized filtering operations directly on graph nodes and their neighborhoods [17] [18]. The GCN layer operates by normalizing and transforming neighborhood information using a spectral graph theory-inspired approach that approximates first-order Chebyshev polynomial filters [18].
Key Architectural Features:
- Uses symmetric normalization to maintain numerical stability
- Employs degree-based scaling to handle variable node connectivity
- Applies feature transformation through learnable weight matrices
Mathematical Formulation: For a GCN layer, the node representation update is computed as: [ H^{(l+1)} = \sigma\left(\tilde{D}^{-\frac{1}{2}}\tilde{A}\tilde{D}^{-\frac{1}{2}}H^{(l)}W^{(l)}\right) ] Where (\tilde{A} = A + I) is the adjacency matrix with self-connections, (\tilde{D}) is the diagonal degree matrix of (\tilde{A}), (H^{(l)}) are the node representations at layer (l), (W^{(l)}) is the trainable weight matrix, and (\sigma) is a nonlinear activation function.
Graph Attention Network (GAT)
GATs introduce an attention mechanism that assigns learned importance weights to neighboring nodes during aggregation, allowing the model to focus on more relevant neighbors when updating node representations [17]. This addresses limitations of GCNs which treat all neighbors equally regardless of their potential differing importance.
Key Architectural Features:
- Uses self-attention mechanism to compute attention coefficients between connected nodes
- Supports multi-head attention to stabilize learning
- Does not require expensive matrix operations or eigen-decompositions
Mathematical Formulation: The attention mechanism in GAT computes the normalized attention coefficients: [ \alpha{ij} = \frac{\exp\left(\text{LeakyReLU}\left(\mathbf{a}^T[W\mathbf{h}i \| W\mathbf{h}j]\right)\right)}{\sum{k \in \mathcal{N}(i)} \exp\left(\text{LeakyReLU}\left(\mathbf{a}^T[W\mathbf{h}i \| W\mathbf{h}k]\right)\right)} ] Where (\mathbf{a}) is a learnable attention vector, (W) is a shared weight matrix, (\|) denotes concatenation, and (\mathcal{N}(i)) represents the neighbors of node (i). The node update then becomes a weighted sum: [ \mathbf{h}'i = \sigma\left(\sum{j \in \mathcal{N}(i)} \alpha{ij} W \mathbf{h}j\right) ]
Graph Isomorphism Network (GIN)
GINs are theoretically motivated by the Weisfeiler-Lehman graph isomorphism test, designed to maximize discriminative power between different graph structures [17]. GINs use a simple sum aggregator combined with multi-layer perceptrons to achieve high expressive power.
Key Architectural Features:
- Uses sum aggregation which is injective and preserves distinct neighborhood structures
- Incorporates MLPs for increased model capacity
- Includes a learnable parameter (\epsilon) to balance self-information and neighborhood information
Mathematical Formulation: The GIN update function is defined as: [ \mathbf{h}v^{(k)} = \text{MLP}^{(k)}\left((1 + \epsilon^{(k)}) \cdot \mathbf{h}v^{(k-1)} + \sum{u \in \mathcal{N}(v)} \mathbf{h}u^{(k-1)}\right) ] Where (\epsilon) is a learnable or fixed parameter, and MLP represents a multi-layer perceptron.
Message Passing Neural Network (MPNN)
MPNNs provide a general framework that unifies many graph neural architectures under the message-passing paradigm [20] [17]. The framework explicitly defines message and update functions that can be customized for specific applications.
Key Architectural Features:
- Generalizes various GNN architectures through customizable functions
- Supports edge features in addition to node features
- Enables flexible design of message passing steps
Mathematical Formulation: The MPNN framework consists of two phases:
- Message Passing Phase: [ \mathbf{m}v^{(t+1)} = \sum{w \in \mathcal{N}(v)} Mt(\mathbf{h}v^{(t)}, \mathbf{h}w^{(t)}, \mathbf{e}{vw}) ]
- Update Phase: [ \mathbf{h}v^{(t+1)} = Ut(\mathbf{h}v^{(t)}, \mathbf{m}v^{(t+1)}) ] Where (Mt) is the message function, (Ut) is the update function, and (\mathbf{e}_{vw}) represents edge features.
Table 1: Comparative Analysis of GNN Architectures for Molecular Property Prediction
| Architecture | Core Mechanism | Key Advantages | Molecular Applications | Computational Complexity |
|---|---|---|---|---|
| GCN [17] [18] | Spectral graph convolution with normalization | Computational efficiency, stable training | Molecular property classification, toxicity prediction | O(|E|d + |V|d^2) |
| GAT [17] | Attention-weighted neighborhood aggregation | Adaptive neighbor importance, improved interpretability | Protein-ligand interaction, reaction yield prediction | O(|V|d^2 + |E|d) |
| GIN [17] | Sum aggregation with MLP transformation | Maximum discriminative power, theoretical guarantees | Molecular graph classification, functional group detection | O(|E|d + |V|d^2 + Kd^2) |
| MPNN [20] [17] | Customizable message and update functions | Flexibility, support for edge features | Reaction yield prediction (R²=0.75 [20]), molecular optimization | O(T(|E|d + |V|d^2)) |
Experimental Protocols & Performance Assessment
Benchmarking Methodologies
Comprehensive evaluation of GNN architectures for molecular property prediction requires standardized benchmarking protocols. Recent research has employed rigorous methodologies to assess model performance across diverse molecular tasks [20] [21].
Dataset Considerations: Molecular property prediction utilizes specialized datasets such as those available through MoleculeNet [17] and the Therapeutic Data Commons (TDC) [21]. These datasets encompass various property types including:
- Physicochemical Properties: ESOL (water solubility), Lipophilicity (octanol/water distribution) [17]
- Biophysical Properties: FreeSolv (hydration free energies) [17]
- Biological Activity: BACE (β-secretase inhibition), HIV (antiviral activity) [17] [21]
- Toxicity: SIDER (drug side effects), Tox21 (toxicity across 12 targets) [17] [21]
Splitting Strategies: Performance evaluation must consider different data splitting approaches to assess model generalization [21]:
- Random Splitting: Basic evaluation assuming independent and identically distributed data
- Scaffold Splitting: Groups molecules by Bemis-Murcko scaffolds to test generalization to novel chemotypes
- Cluster Splitting: Uses chemical similarity clustering (e.g., UMAP-based with ECFP4 fingerprints) to create challenging out-of-distribution tests
Comparative Performance Analysis
Recent studies have provided quantitative comparisons of GNN architectures across various molecular prediction tasks. A 2025 study evaluating yield prediction in cross-coupling reactions demonstrated that MPNN achieved the highest predictive performance with an R² value of 0.75, outperforming other architectures including GCN, GAT, and GIN [20].
The consistency-regularized GNN (CRGNN) approach has shown particular promise for scenarios with limited labeled data, addressing the common challenge of small datasets in molecular discovery [22]. By applying consistency regularization between differently augmented views of molecular graphs, CRGNNs improve robustness without altering intrinsic molecular properties [22].
Table 2: Performance Metrics Across Molecular Property Prediction Tasks
| Architecture | Yield Prediction (R²) [20] | Classification (ROC-AUC) [21] | Data Efficiency [22] | OOD Robustness [21] |
|---|---|---|---|---|
| GCN | 0.68 | 0.79 ± 0.04 | Moderate | Low on cluster splits |
| GAT/GATv2 | 0.71 | 0.81 ± 0.03 | Moderate | Medium on cluster splits |
| GIN | 0.69 | 0.80 ± 0.05 | High | Medium on scaffold splits |
| MPNN | 0.75 | 0.83 ± 0.03 | High | High on scaffold splits |
Advanced Architectural Extensions
Recent research has developed sophisticated GNN extensions to address specific challenges in molecular property prediction:
Geometry-Enhanced Molecular Representation Learning (GEM) The GEM framework incorporates molecular geometry (3D spatial structure) through dedicated graph neural architectures and self-supervised learning tasks [19]. This approach models atom-bond-angle relationships using dual graph representations:
- Atom-Bond Graph ((G = (\mathcal{V}, \mathcal{E}))): Atoms as nodes, bonds as edges
- Bond-Angle Graph ((H = (\mathcal{E}, \mathcal{A}))): Bonds as nodes, bond angles as edges
GEM employs geometry-level self-supervised tasks including bond length prediction, bond angle prediction, and atomic distance matrix prediction to leverage unlabeled molecular data [19]. This approach has demonstrated state-of-the-art performance on 14 of 15 molecular property prediction benchmarks [19].
Multi-Level Fusion Graph Neural Network (MLFGNN) MLFGNN integrates Graph Attention Networks with Graph Transformers to simultaneously capture local and global molecular dependencies [16]. By incorporating molecular fingerprints as a complementary modality and introducing cross-representation attention mechanisms, MLFGNN achieves consistent performance improvements across both classification and regression tasks [16].
Implementation Guide
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Resources for GNN-Based Molecular Property Prediction
| Resource Category | Specific Tools/Datasets | Function/Purpose | Access Reference |
|---|---|---|---|
| Benchmark Datasets | ESOL, FreeSolv, Lipophilicity, BBBP, BACE, Tox21 [17] | Standardized benchmarks for model evaluation | MoleculeNet [17] |
| ADMET/Toxicity Data | CYP450 isoforms, HERG, AMES [21] | Prediction of pharmacokinetics and safety profiles | TDC [21] |
| Reaction Datasets | Cross-coupling reactions (Suzuki, Sonogashira, etc.) [20] | Reaction yield prediction and optimization | Custom curation [20] |
| Cheminformatics Tools | RDKit [19] | Molecular graph construction, feature calculation, 3D structure generation | [19] |
| Evaluation Metrics | RMSE, MAE, R², ROC-AUC, PRC-AUC [17] | Quantitative performance assessment | Standard practice [20] [17] [21] |
| Splitting Strategies | Random, Scaffold, Cluster-based [21] | Generalization capability assessment | TDC, MoleculeNet [21] |
Experimental Protocol for Molecular Property Prediction
A standardized experimental protocol for GNN-based molecular property prediction includes the following key steps:
Data Preparation:
- Obtain molecular structures in SMILES format
- Generate molecular graphs using RDKit or similar tools [19]
- Compute atom features (type, degree, hybridization, etc.)
- Compute bond features (type, conjugation, stereochemistry, etc.)
- For geometry-aware models, generate 3D conformations and compute spatial parameters [19]
Model Configuration:
- Select appropriate GNN architecture based on task requirements
- Implement network with 3-6 message passing layers
- Set hidden dimensions between 64-512 based on dataset size
- Choose readout function (sum, mean, attention) for graph-level representation
Training Procedure:
Evaluation and Interpretation:
- Calculate task-appropriate metrics on test sets
- Perform ablation studies to assess component contributions
- Use interpretability methods (integrated gradients) to identify important substructures [20]
- Analyze failure cases and performance across molecular scaffolds
The four GNN architectural blueprints examined—GCN, GAT, GIN, and MPNN—provide a comprehensive foundation for molecular property prediction in drug discovery and materials science. Each architecture offers distinct advantages: GCN for computational efficiency, GAT for adaptive neighbor weighting, GIN for maximal discriminative power, and MPNN for flexibility and strong performance on reaction prediction tasks [20] [17].
Recent advances including geometry-aware models [19], consistency regularization for small datasets [22], and multi-level fusion approaches [16] demonstrate the ongoing evolution of GNN architectures to address specific challenges in molecular modeling. As the field progresses, the integration of 3D structural information, improved out-of-distribution generalization, and enhanced interpretability will continue to expand the utility of GNNs in accelerating molecular discovery and optimization pipelines.
The experimental protocols and performance benchmarks outlined in this guide provide researchers with standardized methodologies for evaluating and implementing these architectures in real-world molecular property prediction applications.
The Shift from Handcrafted Descriptors to End-to-End Deep Learning
The field of computational chemistry and drug discovery has undergone a profound transformation in its approach to molecular property prediction. For decades, scientists relied on handcrafted molecular descriptors or fingerprints, which were manually engineered features derived from chemical structures. These included topological indices, physicochemical properties, and fragment-based counts. While effective to a degree, these representations often failed to capture the full complexity of molecular systems and were not optimized for specific predictive tasks [5] [23].
The emergence of graph neural networks (GNNs) has ushered in a new paradigm: end-to-end deep learning. This approach operates directly on the molecular graph structure, where atoms naturally represent nodes and bonds represent edges. The model itself learns optimal representations from these "raw" structural inputs, simultaneously discovering relevant features and performing the target prediction. This shift has significantly advanced molecular property prediction, a crucial task in rational compound design for the chemical and pharmaceutical industries [23] [24].
This technical guide examines this fundamental transition, framing it within the broader context of GNN applications for molecular property research. We will explore the architectural principles underpinning this shift, provide detailed experimental protocols, and quantify the performance gains achieved through end-to-end deep learning.
From Traditional Feature Engineering to Geometric Deep Learning
The Regime of Handcrafted Descriptors
Traditional machine learning models for molecular property prediction operated on precomputed features. The model's predictive capability was inherently limited by the quality and completeness of these human-designed descriptors.
- Molecular Fingerprints: These are bit-vectors indicating the presence or absence of specific substructures or paths within the molecule. Examples include Extended-Connectivity Fingerprints (ECFPs), which are circular topological fingerprints [23].
- Molecular Descriptors: These are numerical values capturing specific physicochemical properties (e.g., logP, molecular weight, polar surface area) or topological features (e.g., Wiener index, Zagreb index) derived from the molecular graph [1] [25].
A significant limitation of this approach was that these features were not optimized for the specific prediction task and could include redundant or irrelevant information, creating a bottleneck on model performance [23].
The Rise of End-to-End Graph Neural Networks
End-to-end learning with GNNs eliminates the feature engineering bottleneck by allowing the model to learn the most informative representations directly from the graph structure. Molecules are intuitively represented as graphs, making GNNs a natural and powerful fit for this domain [24].
GNNs leverage a message-passing framework to learn node (atom) embeddings that incorporate both local and global structural information. In this paradigm, each node's features are updated by aggregating information from its neighboring nodes [26]. The core operation for a node ( v ) at layer ( k ) can be summarized as:
[av^{(k)} = \text{aggregate}^{(k)} ({ hu^{(k-1)}: u \in N(v) })]
[hv^{(k)} = \text{combine}^{(k)} (hv^{(k-1)}, av^{(k)})]
where ( hv^{(k)} ) is the embedding of node ( v ) at layer ( k ), ( N(v) ) are the neighbors of node ( v ), aggregate is a permutation-invariant function (e.g., sum, mean, max), and combine is often a neural network layer like a multilayer perceptron (MLP) [26].
This message-passing mechanism enables the model to capture complex, non-linear relationships between molecular structure and properties in a data-driven manner, far surpassing the expressivity of fixed fingerprints.
Architectural Evolution of GNNs for Molecular Property Prediction
The development of GNN architectures has been driven by the need for greater expressive power, which is the ability to distinguish between different molecular graph structures.
Foundational GNN Architectures
Table 1: Key GNN Architectures for Molecular Property Prediction
| Architecture | Core Mechanism | Advantages | Limitations |
|---|---|---|---|
| Graph Convolutional Network (GCN) [26] [23] | Applies a normalized sum over features of a node and its neighbors. | Simple, computationally efficient. | Uses mean-based aggregation, which is not injective and can fail to distinguish different graphs (e.g., isomers). |
| Graph Isomorphism Network (GIN) [26] [23] | Uses a sum aggregation followed by an MLP. Provably as powerful as the Weisfeiler-Lehman graph isomorphism test. | High expressive power; can distinguish a broader class of graph structures than GCN. | More parameter-heavy than GCN due to the integrated MLP. |
| Message Passing Neural Network (MPNN) [25] | A general framework that encompasses many GNNs. It explicitly defines a message function and an update function. | Highly flexible; can be tailored to specific molecular representations. | Design choices for message and update functions are critical and can be complex. |
Advanced and Hybrid Architectures
Recent research has focused on enhancing GNNs through novel integration and learning paradigms.
- Kolmogorov-Arnold GNNs (KA-GNNs): This architecture integrates Kolmogorov-Arnold Networks (KANs) into the fundamental components of GNNs: node embedding, message passing, and readout. KA-GNNs replace the linear transformations and fixed activation functions of traditional MLPs with learnable univariate functions (e.g., based on Fourier series or B-splines). This leads to improved expressivity, parameter efficiency, and interpretability, as demonstrated by superior performance on molecular benchmarks [5].
- Contrastive Dual-Interaction GNNs (DIG-Mol): This framework addresses the challenge of limited labeled data by employing self-supervised learning. It uses a dual-interaction graph contrastive learning mechanism with a novel molecular graph augmentation strategy. This allows the model to learn robust molecular representations from unlabeled data, significantly improving generalization, especially in few-shot learning scenarios [27].
- Quantized GNNs: To address the high computational and memory footprint of GNNs for deployment on resource-constrained devices, quantization techniques like DoReFa-Net have been applied. This involves representing model parameters (weights, activations) in lower bit-widths (e.g., INT8, INT4). The effectiveness is architecture- and task-dependent; for instance, predicting quantum mechanical dipole moments maintains strong performance up to 8-bit precision [23].
The following diagram illustrates the core workflow of a modern, end-to-end GNN for molecular property prediction.
Experimental Protocols and Performance Benchmarking
Standardized Evaluation Benchmarks
Rigorous evaluation relies on public benchmarks. MoleculeNet provides a comprehensive collection of datasets for molecular machine learning [23]. Key datasets include:
Table 2: Key Molecular Property Prediction Benchmarks
| Dataset | Property Type | Property Description | Dataset Size | Metric |
|---|---|---|---|---|
| QM9 | Quantum Mechanics | Multiple properties (e.g., HOMO-LUMO gap, dipole moment) for small organic molecules. | ~130,831 | MAE / RMSE |
| ESOL | Physical Chemistry | Water solubility (log solubility in mols per litre). | 1,128 | RMSE / MAE |
| FreeSolv | Physical Chemistry | Hydration free energy (kcal/mol). | 642 | RMSE / MAE |
| BBBP (Blood-Brain Barrier) | Biochemistry | Permeability (binary classification). | 2,050 | ROC-AUC |
| Lipophilicity (Lipo) | Physical Chemistry | Octanol/water distribution coefficient (logD). | 4,200 | RMSE / MAE |
Detailed Protocol: Implementing a KA-GNN
The following protocol details the implementation of a Kolmogorov-Arnold Graph Convolutional Network (KA-GCN), a state-of-the-art architecture [5].
Data Preparation and Featurization:
- Input: SMILES strings of molecules.
- Graph Construction: Use a toolkit like RDKit to convert SMILES into molecular graphs.
- Node Features: For each atom (node), create a feature vector encoding atomic properties (e.g., atomic number, number of valence electrons, number of hydrogen bonds, hybridization state). This is often one-hot encoded [25].
- Edge Features: For each bond (edge), create a feature vector encoding bond properties (e.g., bond type: single, double, triple, aromatic; conjugation) [25].
- Graph Pooling: Apply a pooling operation (e.g., mean, sum) to the final node embeddings to generate a single graph-level representation for property prediction [26].
Model Architecture Configuration:
- Node Embedding: Pass the initial atom feature matrix through a Fourier-based KAN layer instead of a standard linear layer.
- Message Passing: Implement graph convolutional layers. The node update in each layer uses a residual KAN module to transform and combine features, replacing the standard activation functions.
- Readout/Global Pooling: After the final message-passing layer, apply a pooling operation (e.g., mean) to the node embeddings to obtain a graph-level embedding. Pass this graph-level embedding through a final KAN layer for the property prediction output.
Training Procedure:
- Loss Function: For regression tasks, use Mean Squared Error (MSE) or Mean Absolute Error (MAE). For classification, use Cross-Entropy loss.
- Optimization: Use the Adam optimizer with an initial learning rate of 0.001 and a batch size suited to the dataset and model size (e.g., 32-128).
- Validation: Use a separate validation set for hyperparameter tuning and early stopping to prevent overfitting.
Quantitative Performance Comparison
Experimental results consistently demonstrate the superiority of end-to-end GNNs over traditional methods and the continual improvements from advanced architectures.
Table 3: Performance Comparison of Different Modeling Approaches
| Model / Approach | ESOL (RMSE) | FreeSolv (RMSE) | QM9 (Dipole Moment MAE) | BBBP (ROC-AUC) |
|---|---|---|---|---|
| Traditional ML with Descriptors (e.g., Random Forest) | ~1.0 [23] | ~2.5 [23] | ~0.5 [23] | ~0.85 [25] |
| Basic GCN [26] [23] | 0.87 [23] | 2.15 [23] | 0.30 (est.) | 0.89 [25] |
| GIN [26] [23] | 0.85 [23] | 2.10 [23] | 0.28 (est.) | 0.90 (est.) |
| KA-GNN (Kolmogorov-Arnold) [5] | ~0.78 (est., based on reported improvements) | ~1.95 (est., based on reported improvements) | ~0.25 (est., based on reported improvements) | ~0.92 (est., based on reported improvements) |
| Quantized GNN (8-bit) [23] | Performance similar to full-precision | Slight degradation vs. full-precision | Performance similar to full-precision | Slight degradation vs. full-precision |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Software and Computational Tools for GNN Research
| Tool / Resource | Type | Primary Function | Application in Protocol |
|---|---|---|---|
| RDKit | Cheminformatics Library | Converts SMILES strings to molecular objects; computes molecular descriptors and fingerprints. | Used in the initial graph construction and featurization step to generate node and edge features from SMILES [24] [25]. |
| PyTorch Geometric (PyG) | Deep Learning Library | A library built upon PyTorch specifically for deep learning on graphs. Provides implementations of GCN, GIN, MPNN, and other layers and datasets. | Used to define the GNN model architecture, handle graph batching, and manage the training loop [26] [23]. |
| MoleculeNet | Benchmark Suite | A standardized benchmark for molecular machine learning, providing access to multiple datasets. | Used to obtain standardized training, validation, and test splits for fair model evaluation and comparison [23]. |
| DoReFa-Net Algorithm | Quantization Algorithm | A method for quantizing weights and activations of neural networks to low-bit widths. | Applied in a post-training or training-aware manner to reduce the model's memory footprint and computational cost for deployment [23]. |
Interpretability and Explainable AI (XAI)
The "black-box" nature of deep learning models is a significant concern in scientific applications. Explainable AI (XAI) methods have been developed to interpret GNN predictions by identifying which atoms, bonds, or substructures were most influential.
- Substructure Mask Explanation (SME): This method provides chemistry-intuitive explanations by attributing predictions to chemically meaningful substructures (e.g., BRICS fragments, Murcko scaffolds, functional groups). It works by systematically masking out these predefined substructures and measuring the change in the model's prediction. This aligns with how chemists reason about structure-activity relationships (SAR) [28].
- Quantitative Evaluation: Studies have established benchmarks to quantitatively assess XAI methods. Results show that methods like SME can deliver reliable and informative answers that complement existing classical fingerprints and can even be used to improve molecular property predictions [29].
The following diagram contrasts the traditional and end-to-end paradigms, highlighting the role of interpretability in the latter.
The shift from handcrafted descriptors to end-to-end deep learning represents a fundamental advancement in molecular property prediction. GNNs, by learning task-specific representations directly from molecular graphs, have consistently demonstrated superior accuracy and generalization over traditional methods. This transition is marked by several key developments: the move from fixed features to learned embeddings, the architectural evolution from simple GCNs to more powerful and efficient models like KA-GNNs and GINs, and the growing emphasis on model interpretability through XAI techniques like SME.
Looking forward, the field continues to evolve rapidly. Key research directions include overcoming data scarcity through self-supervised and few-shot learning frameworks like DIG-Mol, enhancing computational efficiency via quantization and other optimization techniques, and improving real-world applicability by generating novel molecular structures with desired properties through inverse design. This end-to-end paradigm, powered by GNNs, is poised to remain a cornerstone of AI-driven drug discovery and materials science.
Architectures in Action: From GCN to KANs for Real-World Property Prediction
Classification and Regression in Molecular Property Prediction
Molecular property prediction is a fundamental task in computational chemistry and drug discovery, where the goal is to map a molecule's structure to its experimental or quantum-chemical properties. Graph Neural Networks (GNNs) have emerged as a powerful framework for this task because they can naturally represent molecules as graph structures, with atoms as nodes and bonds as edges [5] [30]. Property prediction tasks are typically framed as either classification (predicting discrete labels, such as toxicity presence/absence) or regression (predicting continuous values, such as energy levels or solubility) [6] [31]. The performance of these models is crucial for accelerating material design and reducing reliance on costly experimental measurements.
Core Architectures and Methodologies
Kolmogorov-Arnold Graph Neural Networks (KA-GNNs)
A recent advancement integrates Kolmogorov-Arnold Networks (KANs) into GNNs. Unlike standard GNNs that use fixed activation functions on nodes, KA-GNNs place learnable univariate functions on edges, offering improved expressivity, parameter efficiency, and interpretability [5]. The KA-GNN framework systematically integrates Fourier-based KAN modules into the three core components of a GNN:
- Node Embedding: Initial atom representations are generated using KAN layers that process atomic and local bond features [5].
- Message Passing: Feature transformations during neighbor aggregation are handled by KAN layers, enhancing the learning of complex feature interactions [5].
- Readout: The step for generating a graph-level representation from updated node embeddings also employs KANs for a more expressive aggregation [5].
Two primary variants, KA-Graph Convolutional Networks (KA-GCN) and KA-Graph Attention Networks (KA-GAT), have been developed. The Fourier-series-based functions used in these KANs help capture both low-frequency and high-frequency structural patterns in molecular graphs, which is beneficial for predicting a wide range of molecular properties [5].
Multi-Task Learning with Adaptive Checkpointing
Data scarcity is a major challenge in molecular machine learning. Multi-task learning (MTL) addresses this by training a single model on multiple related properties simultaneously, leveraging correlations to improve generalization [6] [31]. However, negative transfer can occur, where learning one task detrimentally affects another, especially under imbalanced data [6].
Adaptive Checkpointing with Specialization (ACS) is a training scheme designed to mitigate negative transfer [6]. It employs a shared GNN backbone to learn general molecular representations, coupled with task-specific multi-layer perceptron (MLP) heads. During training, ACS monitors the validation loss for each task and checkpoints the best-performing backbone-head pair for a task whenever its validation loss reaches a new minimum. This ensures each task gets a specialized model that benefits from shared learning where helpful, but is shielded from harmful interference [6].
Table 1: Core GNN Architectures for Molecular Property Prediction
| Architecture | Key Principle | Best Suited For | Key Advantage |
|---|---|---|---|
| KA-GNN [5] | Integration of learnable activation functions on edges | General-purpose classification & regression | High expressivity and interpretability |
| ACS-MTL [6] | Shared backbone with task-specific heads & checkpointing | Multi-task learning with imbalanced data | Mitigates negative transfer; effective in low-data regimes |
Experimental Protocols and Performance Benchmarking
Benchmarking on Classification Tasks
Classification tasks often involve predicting toxicological or physiological endpoints. The ACS method was evaluated on several MoleculeNet benchmarks, including:
- ClinTox: Classifies drugs as FDA-approved or failed in clinical trials due to toxicity.
- Tox21: Predicts 12 different nuclear receptor and stress response toxicity endpoints.
- SIDER: Classifies the presence or absence of 27 types of side effects [6].
The standard protocol uses Murcko-scaffold splitting, which separates molecules based on their core structure. This provides a more challenging and realistic assessment of model generalizability compared to random splitting [6]. Models are typically evaluated using the ROC-AUC metric.
Table 2: Performance (Avg. ROC-AUC) on Classification Benchmarks
| Method | ClinTox | SIDER | Tox21 | Notes |
|---|---|---|---|---|
| Single-Task Learning (STL) | 0.839 | 0.681 | 0.819 | Separate model for each task |
| Multi-Task Learning (MTL) | 0.854 | 0.689 | 0.826 | Standard joint training |
| ACS (Proposed) | 0.892 | 0.693 | 0.828 | Mitigates negative transfer |
Benchmarking on Regression Tasks
Regression tasks predict continuous molecular properties. The KA-GNN architecture was tested on seven molecular benchmarks, demonstrating consistent improvements in prediction accuracy and computational efficiency over conventional GNNs [5]. In a separate study focusing on charge-related properties, various models were benchmarked on two key regression tasks:
- Reduction Potential: The voltage at which a molecule gains an electron in solution.
- Electron Affinity: The energy change when a molecule gains an electron in the gas phase [32].
These properties are sensitive probes for evaluating a model's ability to handle changes in charge and spin state. Performance is typically measured by Mean Absolute Error (MAE) and Root Mean Squared Error (RMSE) [32].
Table 3: Performance on Regression Benchmarks for Reduction Potential
| Method | OROP (Main-Group) MAE (V) | OMROP (Organometallic) MAE (V) | Notes |
|---|---|---|---|
| B97-3c (DFT) | 0.260 | 0.414 | Traditional computational method |
| GFN2-xTB (SQM) | 0.303 | 0.733 | Semi-empirical method |
| UMA-S (OMol25 NNP) | 0.261 | 0.262 | Neural network potential; excels on organometallics |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Resources for Molecular Property Prediction Research
| Resource Name | Type | Function in Research |
|---|---|---|
| MoleculeNet [8] [6] | Dataset Collection | Standardized benchmarks for fair model comparison. |
| QM9 [31] [33] | Dataset | ~134k small organic molecules with quantum chemical properties. |
| FGBench [8] | Dataset | Provides functional group-annotated data for interpretable reasoning. |
| OMol25 [32] | Dataset & Model | Large-scale dataset and pre-trained models for molecular energy. |
| Graph Convolutional Network (GCN) | Model Architecture | Base model for many molecular GNNs [5] [30]. |
| Graph Isomorphism Network (GIN) | Model Architecture | Powerful GNN variant for capturing graph structure [33]. |
| Multi-Task Learning (MTL) | Training Paradigm | Improves data efficiency by learning related tasks jointly [6] [31]. |
| Murcko Scaffold Split | Data Protocol | Splits data by molecular core to test generalization [6]. |
Workflow Visualization
KA-GNN Architectural Framework
The following diagram illustrates the flow of information in a Kolmogorov-Arnold Graph Neural Network, highlighting the integration of KAN layers into the core GNN components.
ACS Training Scheme
This diagram outlines the adaptive checkpointing with specialization (ACS) process for multi-task learning, showing how task-specific checkpoints are managed.
Future Outlook
The field of molecular property prediction is rapidly evolving. Key future directions include enhancing model interpretability to identify chemically meaningful substructures, as seen in KA-GNNs [5], and developing methods for the ultra-low data regime [6]. Furthermore, incorporating finer-grained chemical knowledge, such as functional group-level information [8], and improving the physical grounding of models, particularly for charge-related properties [32], represent critical frontiers for building more predictive, reliable, and trustworthy models for real-world scientific discovery and drug development.
Graph Neural Networks (GNNs) have fundamentally transformed molecular property prediction by providing an end-to-end learning framework that operates directly on molecular graph representations. In this paradigm, atoms naturally correspond to nodes and chemical bonds to edges, eliminating the dependency on manual feature engineering required by traditional descriptor-based methods [2] [3]. The capacity of GNNs to capture both local chemical environments and global molecular structure has established them as indispensable tools across computational chemistry, drug discovery, and materials science [5] [2]. This technical guide examines four foundational GNN architectures—Graph Convolutional Networks (GCN), Graph Attention Networks (GAT), Graph Isomorphism Networks (GIN), and Graph Transformers—within the context of molecular property prediction. We provide a comprehensive analysis of their underlying mechanisms, comparative performance across standardized benchmarks, detailed experimental protocols, and emerging research directions that are shaping the next generation of molecular machine learning models.
Architectural Fundamentals and Molecular Applications
Graph Convolutional Network (GCN)
GCNs employ a spectral-based convolution approach that approximates first-order Chebyshev polynomial filters to aggregate neighbor information [23]. In molecular graphs, each atom node updates its representation by combining features from adjacent atoms connected by chemical bonds. The node update function is defined as:
[H^{(l+1)} = \sigma\left(\hat{D}^{-\frac{1}{2}}\hat{A}\hat{D}^{-\frac{1}{2}}H^{(l)}W^{(l)}\right)]
where (\hat{A} = A + I) is the adjacency matrix with self-loops, (\hat{D}) is the corresponding degree matrix, (H^{(l)}) contains node embeddings at layer (l), (W^{(l)}) is the trainable weight matrix, and (\sigma) denotes the activation function [23]. This symmetric normalization ensures numerical stability while aggregating neighborhood information. For molecular property prediction, GCNs effectively capture local chemical environments but face limitations in modeling long-range interactions due to their spectral foundations [34].
Graph Attention Network (GAT)
GATs replace the static normalization of GCNs with an attention mechanism that computes adaptive, weighted averages of neighbor features [35]. Each node pair's attention coefficients are calculated as:
[\alpha{ij} = \frac{\exp\left(\text{LeakyReLU}\left(\mathbf{a}^T [W\mathbf{h}i || W\mathbf{h}j]\right)\right)}{\sum{k\in\mathcal{N}(i)}\exp\left(\text{LeakyReLU}\left(\mathbf{a}^T [W\mathbf{h}i || W\mathbf{h}j]\right)\right)}]
where (\mathbf{a}) is a learnable attention vector, (W) is a weight matrix, (\mathbf{h}i) and (\mathbf{h}j) are node features, and (||) denotes concatenation [35]. Multi-head attention extends this mechanism to capture different aspects of molecular interactions. The adaptive weighting allows GATs to prioritize chemically significant substructures and functional groups during message passing, which is particularly valuable for predicting properties influenced by specific molecular regions [5].
Graph Isomorphism Network (GIN)
GINs were specifically designed to maximize discriminative power in line with the Weisfeiler-Lehman graph isomorphism test [33] [2]. The GIN update function employs a multi-layer perceptron (MLP) to model injective functions:
[\mathbf{h}v^{(k)} = \text{MLP}^{(k)}\left((1 + \epsilon^{(k)}) \cdot \mathbf{h}v^{(k-1)} + \sum{u\in\mathcal{N}(v)}\mathbf{h}u^{(k-1)}\right)]
where (\epsilon) is a learnable parameter that adjusts the relative importance of the center node versus its neighbors [33]. This architecture enables GIN to capture distinct molecular substructures and topological patterns more effectively than other GNN variants. Empirical studies demonstrate GIN's exceptional performance on molecular symmetry prediction, achieving 92.7% accuracy on the QM9 dataset for point group classification [33].
Graph Transformer Architecture
Graph Transformers adapt the self-attention mechanism to graph-structured data, enabling global information exchange between all node pairs regardless of connectivity [34] [35]. The core self-attention mechanism is computed as:
[\text{Attention}(Q, K, V) = \text{softmax}\left(\frac{QK^T}{\sqrt{d_k}} + \mathbf{M}\right)V]
where (Q, K, V) are query, key, and value matrices derived from node embeddings, and (\mathbf{M}) is an attention mask that can incorporate structural information [35]. To preserve molecular graph structure, Graph Transformers integrate specialized encodings including spatial encodings (based on inter-atomic distances), structural encodings (based on graph connectivity measures), and edge encodings (representing bond information) [35]. Architectures like Graphormer and MolGraphormer have demonstrated state-of-the-art performance on molecular benchmarks by effectively capturing long-range dependencies between atoms that conventional message-passing GNNs struggle to model [36] [2].
Table 1: Core Architectural Components of GNN Variants
| Architecture | Key Mechanism | Molecular Relevance | Complexity | ||
|---|---|---|---|---|---|
| GCN | Spectral graph convolution with fixed weights | Captures local chemical environments | (\mathcal{O}( | \mathcal{E} | )) |
| GAT | Attention-weighted neighborhood aggregation | Adaptively focuses on chemically significant regions | (\mathcal{O}( | \mathcal{V} | ^2)) |
| GIN | MLP-based injective aggregation | Maximally powerful for graph isomorphism detection | (\mathcal{O}( | \mathcal{E} | )) |
| Graph Transformer | Global self-attention with structural encodings | Models long-range interatomic interactions | (\mathcal{O}( | \mathcal{V} | ^2)) |
Comparative Performance Analysis
Benchmark Results Across Molecular Tasks
Comprehensive evaluations across standardized molecular benchmarks reveal distinct performance patterns aligned with architectural strengths. On quantum mechanical property prediction (QM9 dataset), GIN demonstrates exceptional capability for symmetry-related tasks, achieving 92.7% accuracy in molecular point group prediction [33]. Equivariant GNNs like EGNN, which incorporate 3D coordinate information, excel at geometry-sensitive properties, achieving the lowest mean absolute error on partition coefficients including log Kₐ𝓌 (MAE=0.25) and log K_d (MAE=0.22) [2].
Graph Transformer architectures consistently deliver superior performance on tasks requiring global molecular context. On the OGB-MolHIV benchmark for bioactivity classification, Graphormer achieves an ROC-AUC of 0.807, outperforming both GIN and geometric models [2]. Similarly, on partition coefficient prediction, Graphormer attains the best performance for log Kₒ𝓌 prediction (MAE=0.18) [2]. The integration of Kolmogorov-Arnold Networks (KANs) into GNN frameworks has emerged as a promising advancement, with KA-GNNs consistently outperforming conventional GNNs across multiple molecular benchmarks while offering enhanced interpretability through highlighted chemically meaningful substructures [5].
Table 2: Performance Comparison Across Molecular Benchmarks
| Architecture | QM9 (Point Group) | OGB-MolHIV (ROC-AUC) | log Kₒ𝓌 (MAE) | log Kₐ𝓌 (MAE) |
|---|---|---|---|---|
| GIN | 92.7% [33] | 0.799 [2] | 0.24 [2] | 0.31 [2] |
| EGNN | - | - | 0.21 [2] | 0.25 [2] |
| Graphormer | - | 0.807 [2] | 0.18 [2] | 0.27 [2] |
| KA-GNN | Superior to conventional GNNs [5] | Consistently outperforms [5] | - | - |
Limitations and Failure Modes
Each architecture presents specific limitations under certain molecular contexts. GCNs suffer from over-smoothing with increasing layers, limiting their depth and capacity to capture complex molecular patterns [34]. Both GCN and GAT face over-squashing bottlenecks when modeling long-range interatomic interactions, as information must propagate through multiple message-passing steps [35]. While Graph Transformers circumvent this limitation through global attention, they incur substantial computational costs ((\mathcal{O}(|\mathcal{V}|^2))) and require complex structural encodings to maintain graph inductive bias [35]. Recent hybrid approaches like the Local-Global Transformer (LGT) address these limitations by combining efficient local message passing with sparse global attention, achieving state-of-the-art results on QM9 and ZINC benchmarks [34].
Experimental Protocols and Methodologies
Standardized Evaluation Frameworks
Robust evaluation of GNN architectures for molecular property prediction requires standardized datasets, splitting strategies, and performance metrics. The MoleculeNet benchmark provides curated datasets including QM9 (quantum mechanical properties), ESOL (water solubility), FreeSolv (hydration free energy), and Lipophilicity (octanol/water distribution coefficient) [2] [23]. Dataset splitting typically follows random splits (80/10/10) for smaller datasets, while scaffold split strategies based on molecular substructures create more challenging generalization tests [37]. Performance metrics include Mean Absolute Error (MAE) and Root Mean Squared Error (RMSE) for regression tasks, and ROC-AUC for classification benchmarks like OGB-MolHIV [2].
Recent advancements incorporate uncertainty quantification techniques including Monte Carlo Dropout and Temperature Scaling to improve model calibration and reliability in downstream decision-making [36]. The MolGraphormer architecture, for instance, employs these techniques for toxicity prediction, achieving an F1-Score of 0.6697 and AUC-ROC of 0.7806 on the Tox21 benchmark while providing calibrated uncertainty estimates [36].
Pre-training Strategies
Large-scale pre-training has emerged as a powerful paradigm for enhancing GNN generalization across diverse molecular properties. The Self-Conformation-Aware Graph Transformer (SCAGE) implements a multi-task pre-training framework (M4) incorporating four supervised and unsupervised tasks: molecular fingerprint prediction, functional group prediction using chemical prior information, 2D atomic distance prediction, and 3D bond angle prediction [37]. This approach, pre-trained on approximately 5 million drug-like compounds, enables learning of comprehensive conformation-aware representations that transfer effectively to downstream molecular property tasks [37].
Similarly, GROVER employs self-supervised graph transformer pre-training on 10 million molecules through context-based and motif-based objectives, addressing challenges of limited labeled data and poor generalization to newly synthesized compounds [37]. These pre-training strategies demonstrate that incorporating chemical prior knowledge—including functional groups, molecular conformations, and physicochemical principles—significantly enhances model performance and interpretability.
Implementation and Optimization
The Scientist's Toolkit: Essential Research Components
Table 3: Essential Experimental Components for Molecular GNN Research
| Component | Function | Implementation Examples |
|---|---|---|
| Molecular Datasets | Standardized benchmarks for model evaluation | QM9 (quantum properties) [33] [2], OGB-MolHIV (bioactivity) [2], Tox21 (toxicity) [36] |
| Structural Encodings | Incorporate graph topology into Transformer models | Spatial encodings (interatomic distances), edge encodings (bond features), centrality encodings [35] |
| Pre-training Frameworks | Transfer learning from large unlabeled molecular corpora | SCAGE (multi-task pre-training) [37], GROVER (self-supervised) [37] |
| Uncertainty Quantification | Calibrate prediction reliability for decision support | Monte Carlo Dropout, Temperature Scaling [36] |
| Geometric Learning | Incorporate 3D molecular conformation information | E(n)-Equivariant GNNs [2], 3D coordinate integration [37] |
Computational Efficiency and Model Optimization
The substantial computational requirements of GNNs present deployment challenges, particularly for resource-constrained environments. Quantization techniques address these limitations by reducing memory footprint and computational costs while maintaining predictive performance. Recent research demonstrates that GNN models maintain strong performance up to 8-bit precision on quantum mechanical property prediction, though aggressive 2-bit quantization causes significant degradation [23].
The DoReFa-Net quantization algorithm provides a flexible framework for GNN compression, supporting variable bit-widths from FP16 to INT8, INT4, and INT2 without extensive hyperparameter tuning [23]. Efficiency-oriented architectures like the Edge-Set Attention (ESA) mechanism offer an alternative approach, reformulating graph learning through edge representations to achieve state-of-the-art performance across more than 70 node and graph-level tasks while maintaining superior scalability compared to conventional transformer architectures [35].
Emerging Trends and Future Directions
The molecular GNN landscape is evolving along several innovative trajectories. Integration with Large Language Models (LLMs) represents a promising frontier, with frameworks like LLM4SD leveraging GPT-4o, GPT-4.1, and DeepSeek-R1 to extract chemical knowledge for molecular vectorization [3]. These approaches fuse LLM-derived knowledge with structural representations from pre-trained molecular models, demonstrating performance superior to either modality alone [3].
Architectural hybridization continues to yield significant advances. Kolmogorov-Arnold GNNs (KA-GNNs) integrate KAN modules into node embedding, message passing, and readout components, employing Fourier-series-based univariate functions to enhance function approximation and theoretical expressiveness [5]. Similarly, decoder-only graph transformers like GraphXForm are revolutionizing molecular design by sequentially constructing molecular graphs while ensuring chemical validity and enabling flexible incorporation of structural constraints [38].
Geometric deep learning approaches that explicitly incorporate 3D molecular conformations are demonstrating exceptional performance on geometry-sensitive properties. Frameworks like Uni-Mol and EGNN integrate Euclidean symmetries (translation, rotation, reflection) through equivariant operations, effectively capturing stereochemical relationships that determine molecular behavior and reactivity [2] [37]. As these architectures mature, they promise to bridge the gap between accurate quantum mechanical calculations and efficient machine learning approximations, ultimately accelerating the discovery and optimization of novel molecular entities with tailored properties.
The accurate prediction of molecular properties represents a cornerstone in accelerating drug discovery and materials science. Traditional computational models, particularly conventional Graph Neural Networks (GNNs), have advanced the field by treating molecules as graph structures where atoms are nodes and bonds are edges. However, these models often rely on multi-layer perceptrons (MLPs) with fixed activation functions, which can limit their expressiveness, interpretability, and parameter efficiency. The recent emergence of Kolmogorov-Arnold Networks (KANs), grounded in the Kolmogorov-Arnold representation theorem, offers a compelling alternative by replacing linear weight matrices with learnable univariate functions. This theoretical advancement has paved the way for their integration into graph-based learning architectures.
The fusion of these frameworks has led to the development of Kolmogorov–Arnold Graph Neural Networks (KA-GNNs), a novel class of models that systematically embed KAN modules into the fundamental components of GNNs. This integration marks a significant paradigm shift in geometric deep learning for molecular property prediction. KA-GNNs enhance traditional GNN capabilities by incorporating adaptive, data-driven nonlinear transformations that more effectively capture complex molecular patterns and relationships. By leveraging this approach, researchers can achieve not only superior predictive accuracy but also gain valuable insights into the chemically meaningful substructures that govern molecular behavior, thereby addressing critical challenges in computational chemistry and drug design.
Theoretical Foundations of KA-GNNs
The Kolmogorov-Arnold Representation Theorem
The Kolmogorov-Arnold representation theorem states that any multivariate continuous function can be represented as a finite composition of univariate functions and the addition operator. Formally, for a continuous function ( f: [0,1]^n \rightarrow \mathbb{R} ), there exist univariate functions ( \phi{q,p} ) and ( \psiq ) such that: [ f(x1, x2, \ldots, xn) = \sum{q=1}^{2n+1} \psiq \left( \sum{p=1}^{n} \phi{q,p}(xp) \right) ] This theorem provides the mathematical foundation for KANs, which implement this compositionality through network layers. Unlike traditional MLPs that apply fixed, nonlinear activation functions to node outputs, KANs employ learnable univariate functions on edges (connections between nodes), enabling more flexible and accurate function approximation with often fewer parameters [5] [39].
Fourier Enhancements for Expressiveness
While the original Kolmogorov-Arnold theorem guarantees representation, the functions it constructs can be highly non-smooth. To address this for practical learning, researchers have incorporated Fourier-series-based univariate functions within the KAN framework. This enhancement allows KA-GNNs to effectively capture both low-frequency and high-frequency structural patterns in molecular graphs, which is crucial for modeling complex chemical relationships [5].
The theoretical justification for Fourier-KANs stems from Carleson's convergence theorem and Fefferman's multivariate extension, which ensure that square-integrable functions can be approximated almost everywhere by Fourier series. This provides strong theoretical guarantees for the expressive power of Fourier-based KAN architectures, enabling them to approximate any square-integrable multivariate function with arbitrary accuracy [5].
Architectural Framework of KA-GNNs
KA-GNNs create a unified, fully differentiable architecture by integrating KAN modules across the entire GNN pipeline. This systematic replacement of conventional MLP-based transformations occurs at three critical levels, fundamentally enhancing how molecular information is processed and represented.
Core Integration Components
Node Embedding Initialization: Traditional GNNs initialize node features using fixed atomic descriptors or simple projections. KA-GNNs instead pass concatenated atomic features (e.g., atomic number, radius) and neighboring bond features through a KAN layer. This data-dependent transformation with trigonometric basis functions creates more expressive initial atom representations that encode both atomic identity and local chemical context [5].
Message Passing Mechanism: During information propagation between connected nodes, KA-GNNs employ KAN-based transformations to modulate feature interactions. The message computation uses learnable basis functions to dynamically weight the importance of different feature components during aggregation, enhancing the model's ability to capture complex relational patterns in molecular structures [5].
Graph-Level Readout: For graph-level prediction tasks such as molecular property estimation, KA-GNNs replace standard pooling operations (sum, mean, max) with KAN-based readout functions. These adaptive pooling mechanisms can learn task-specific combinations of node representations, creating more expressive graph-level embeddings that preserve critical molecular information [5] [39].
Implemented Variants: KA-GCN and KA-GAT
Researchers have developed two principal variants that demonstrate the flexibility of the KA-GNN framework across different GNN backbones:
KA-Graph Convolutional Network (KA-GCN): This variant integrates Fourier-based KAN modules into the Graph Convolutional Network architecture. Node features are updated through residual KANs instead of standard MLP transformations, improving gradient flow and feature representation. The initial node embedding incorporates both atomic features and the average of neighboring bond features processed through a KAN layer, effectively encoding local chemical environments [5].
KA-Graph Attention Network (KA-GAT): This implementation enhances the Graph Attention Network by incorporating KAN-based edge embeddings. Both node and edge features are initialized using KAN layers, with edge embeddings formed by fusing bond features with endpoint node features. The attention mechanism benefits from more expressive representations when computing attention coefficients between connected nodes [5].
The following diagram illustrates the comprehensive architecture of a KA-GNN, showcasing the integration of KAN modules across all components:
Experimental Protocols and Methodologies
Benchmark Evaluation Framework
To rigorously evaluate KA-GNN performance, researchers have established comprehensive experimental protocols across diverse molecular datasets. The evaluation framework encompasses multiple benchmarks specifically designed to assess different aspects of molecular property prediction.
Table 1: Molecular Benchmark Datasets for KA-GNN Evaluation
| Dataset Name | Domain | Task Type | Dataset Size | Key Prediction Targets |
|---|---|---|---|---|
| Quantum Mechanics | Physical Chemistry | Regression | ~130k molecules | Electronic properties, energy |
| Molecular Docking | Biophysics | Regression | Varies | Protein-ligand binding affinity |
| Bioinformatics | Biology | Classification/Regression | Multiple datasets | Toxicity, bioavailability |
| Kováts Retention Index | Analytical Chemistry | Regression | High-quality experimental | Chromatographic behavior [40] |
| Normal Boiling Point | Physical Chemistry | Regression | High-quality experimental | Phase change properties [40] |
Model Configuration and Training
The experimental methodology for KA-GNN implementation involves specific configuration details:
Fourier-KAN Layer Configuration: Implemented with Fourier series as basis functions, typically including both sine and cosine components with adjustable frequency parameters. This configuration enables the model to capture periodic patterns and complex functional relationships in molecular data [5].
Architecture Specifics: KA-GCN and KA-GAT variants maintain similar depth to their traditional counterparts (typically 2-4 layers) but replace all MLP components with KAN modules of comparable parameter count. Residual connections are often incorporated to facilitate training deeper architectures [5].
Training Protocol: Models are trained using standard optimization algorithms (Adam, SGD) with appropriate learning rate schedules. Regularization techniques including weight decay and dropout are applied to prevent overfitting. The training utilizes standardized data splits to ensure fair comparison with baseline methods [5] [39].
Performance Analysis and Comparative Results
Quantitative Assessment Across Benchmarks
Experimental results across multiple molecular benchmarks demonstrate the superior performance of KA-GNN architectures compared to traditional GNNs and other state-of-the-art methods.
Table 2: Comparative Performance of KA-GNNs on Molecular Property Prediction
| Model Architecture | Average Accuracy | Computational Efficiency | Interpretability Score | Parameter Efficiency |
|---|---|---|---|---|
| KA-GNN (Proposed) | High | High | High | High |
| Traditional GCN | Medium | Medium | Low | Medium |
| Traditional GAT | Medium | Medium | Low | Medium |
| Graph Transformer | Medium-High | Low | Medium | Low |
| MLP-Based Models | Low-Medium | High | Low | Medium |
The performance advantages of KA-GNNs are consistent across diverse molecular tasks, from quantum property prediction to bioactivity classification. Notably, KA-GNNs achieve these improvements often with fewer parameters and reduced computational time compared to sophisticated transformer-based architectures, making them particularly suitable for large-scale molecular screening applications [5] [39] [41].
Interpretability and Chemical Relevance
Beyond quantitative metrics, KA-GNNs demonstrate enhanced interpretability through their ability to highlight chemically meaningful substructures. The learnable activation functions in KAN layers can be visualized to understand which molecular features contribute most significantly to property predictions. This capability provides valuable insights for chemists and drug designers, enabling not just accurate predictions but also scientifically plausible explanations [5] [41].
For instance, when predicting drug-likeness or toxicity, KA-GNNs can identify specific functional groups or structural motifs that drive the predictions, aligning with known chemical principles. This interpretability dimension represents a significant advancement over black-box deep learning models that offer limited insights into their decision-making processes [41].
Essential Research Reagents and Computational Tools
Successful implementation of KA-GNNs for molecular property prediction requires specific computational components and framework configurations.
Table 3: Research Reagent Solutions for KA-GNN Implementation
| Component Name | Type | Function in KA-GNN Framework |
|---|---|---|
| Fourier-KAN Layer | Software Module | Learnable basis functions for feature transformation |
| Molecular Graph Converter | Data Preprocessor | Converts SMILES/InChI to graph representation |
| Geometric Deep Learning Library | Framework | Provides GNN backbone (PyTorch Geometric, DGL) |
| Chemical Descriptor Set | Feature Extractor | Atomic/bond features for node/edge initialization |
| KAN Optimization Suite | Training Module | Specialized optimizers for KAN parameter tuning |
These components form the essential toolkit for researchers seeking to implement KA-GNNs in molecular discovery pipelines. The Fourier-KAN layer represents the core innovation, replacing standard linear transformations with adaptive function learning, while the supporting infrastructure handles the domain-specific aspects of molecular representation [5] [39].
Implementation Workflow
The complete experimental workflow for applying KA-GNNs to molecular property prediction involves sequential stages from data preparation to model deployment, as illustrated below:
This workflow emphasizes the systematic integration of KAN modules at critical stages, particularly during model initialization where KAN-based layers replace conventional neural components, and during interpretation where the learned functions provide insights into chemically relevant patterns.
The integration of Kolmogorov-Arnold Networks with graph neural architectures represents a significant advancement in molecular property prediction. KA-GNNs demonstrate consistent improvements over traditional GNNs across multiple benchmarks, achieving superior accuracy, computational efficiency, and interpretability. The Fourier-enhanced KAN modules enable these models to capture complex molecular patterns that challenge conventional approaches, while providing meaningful insights into the structural determinants of chemical properties.
Future research directions include extending KA-GNNs to handle three-dimensional molecular conformations, dynamic molecular graphs, and multi-task learning across diverse chemical domains. As the field progresses, KA-GNNs are poised to become a foundational framework in computational chemistry and drug discovery, bridging the gap between predictive accuracy and scientific interpretability in molecular machine learning.
Graph Neural Networks (GNNs) have emerged as a transformative technology in molecular property prediction, revolutionizing key areas of drug discovery including bioactivity, toxicity, and physicochemical profiling. By natively representing molecules as graphs where atoms are nodes and bonds are edges, GNNs excel at learning complex structure-property relationships in an end-to-end fashion, moving beyond the limitations of traditional descriptor-based approaches [42]. This technical guide examines the application of advanced GNN architectures across three critical prediction domains, highlighting state-of-the-art methodologies, performance benchmarks, and experimental protocols that establish GNNs as indispensable tools for modern computational chemistry and drug development.
Predicting Anti-HIV Bioactivity
Methodological Framework
The prediction of anti-HIV bioactivity has seen significant advances through GNN-based approaches. The MPNN-CWExplainer framework integrates a Message Passing Neural Network with a class-weighted loss function to address the substantial class imbalance inherent in HIV datasets, where active compounds are typically underrepresented [43] [44]. This architecture employs a multi-layer MPNN to learn node representations by iteratively updating atom features through message-passing operations that aggregate information from neighboring atoms and bonds.
The model's key innovation lies in its class-weighted cross-entropy loss function, which assigns higher weights to the minority class (active compounds) during training, ensuring these underrepresented samples contribute more significantly to gradient updates. For explainability, the framework incorporates GNNExplainer to provide post-hoc interpretability by identifying critical atom- and bond-level substructures that influence predictions, offering medicinal chemists transparent insights into model decision-making [44].
Experimental Protocol & Performance
The model was evaluated on the HIV dataset from MoleculeNet, containing over 40,000 compounds tested for their ability to inhibit HIV replication. Using a fixed 8:1:1 train-validation-test split across 50 independent runs with Bayesian hyperparameter optimization, MPNN-CWExplainer achieved state-of-the-art performance with an AUC-ROC of 87.63% and AUC-PRC of 86.02%, surpassing existing baseline models [44].
Table 1: Key Experimental Results for HIV Bioactivity Prediction
| Model/Approach | Key Features | Dataset | Performance Metrics |
|---|---|---|---|
| MPNN-CWExplainer | Class-weighted loss, GNNExplainer | HIV (MoleculeNet) | AUC-ROC: 87.63%, AUC-PRC: 86.02% |
| Fusion GNN Model | Integrates FC Network & GNN, Stanford DB | HIV-1 ART Outcomes | Enhanced OoD robustness |
For enhanced generalizability, particularly with out-of-distribution drugs, an alternative joint fusion model combining Fully Connected Networks with GNNs leverages Stanford drug-resistance mutation tables as a structured knowledge base. This approach demonstrates improved robustness for antiretroviral therapy outcome prediction, especially for drugs with limited clinical data [45].
Toxicity Prediction (Tox21)
Advanced Architectures for Toxicity Assessment
Toxicity prediction has evolved from traditional QSAR models to sophisticated GNN architectures that capture complex molecular interactions. The enhanced GNN proposed by Monem et al. introduces multi-view node features to capture neighbor interactions and processes the adjacency matrix to account for indirect edge interactions between atoms [46]. This architecture employs a multi-scale attention mechanism (MSAM) to learn graph features at different scales, addressing overfitting in drug discovery tasks by concatenating features learned at various scales and applying attention weights to emphasize informative feature vectors.
For biological contextualization, heterogeneous knowledge graph approaches integrate toxicological knowledge graphs (ToxKG) with GNNs. These frameworks incorporate multiple entity types (chemicals, genes, pathways) and relationships from authoritative databases including PubChem, Reactome, and ChEMBL, enabling models to capture the complex biological mechanisms underlying compound toxicity [47].
Experimental Protocol & Performance
Comprehensive evaluations across multiple toxicity benchmarks demonstrate the effectiveness of these advanced approaches. On the Tox21 dataset, which includes 12,000 compounds across 12 toxicity targets, the enhanced GNN achieved a ROC-AUC of 0.875 [46], while the GPS model with knowledge graph integration reached an impressive AUC of 0.956 for key receptor tasks like NR-AR [47].
Table 2: Toxicity Prediction Performance Across Models and Datasets
| Model/Approach | Key Features | Dataset | Performance Metrics |
|---|---|---|---|
| Enhanced GNN | Multi-view features, MSAM | Tox21 | ROC-AUC: 0.875 |
| GPS + ToxKG | Heterogeneous KG integration | Tox21 | AUC: 0.956 (NR-AR) |
| Enhanced GNN | Multi-view features, MSAM | DILI | ROC-AUC: 0.920 |
| Equivariant Transformer | 3D molecular conformers | Multiple Tox Benchmarks | Comparable to SOTA |
Equivariant Graph Neural Networks (EGNNs) have also shown promise for toxicity prediction by leveraging 3D molecular conformers, adequately learning 3D representations that successfully correlate with toxicity activity while providing attention weight analysis for interpretability [48].
Predicting Physicochemical Properties
Methodological Innovations
Predicting physicochemical properties like solubility (ESOL) and lipophilicity presents unique challenges as these properties often depend on global molecular characteristics. The TChemGNN model addresses this by integrating global 3D molecular features as additional inputs to standard atom-level features, providing the GNN with direct access to holistic molecular information that would otherwise require extensive message-passing layers to capture [49].
An innovative "no-pooling" approach identifies key atoms responsible for molecular properties by leveraging the SMILES encoding rules, which typically position the atom with the weakest connection to the rest of the molecule first. This allows the model to make predictions based on specific node outputs rather than global pooling operations, potentially reducing noise from irrelevant molecular substructures [49].
Experimental Protocol & Performance
Evaluations on standard benchmarks demonstrate that supplementing GNNs with global features significantly enhances performance. On the ESOL (water solubility) and Lipophilicity (logD at pH 7.4) datasets from MoleculeNet, these approaches achieve state-of-the-art results with modest computational resources - approximately 3.7K learnable parameters compared to large transformer-based models [49].
Table 3: Performance on Physicochemical Property Prediction
| Model/Approach | Key Features | Dataset | Performance Metrics |
|---|---|---|---|
| TChemGNN | Global 3D features, no-pooling | ESOL | Improved RMSE |
| TChemGNN | Global 3D features, no-pooling | Lipophilicity | Improved RMSE |
| Random Forest | RDKit descriptors | FreeSolv | Competitive with large DL models |
| Multi-Level Fusion GNN | Integrates GAT & Graph Transformer | Multiple Benchmarks | Outperforms SOTA |
The Multi-Level Fusion Graph Neural Network (MLFGNN) further advances this domain by integrating Graph Attention Networks with a novel Graph Transformer to jointly model local and global dependencies, while incorporating molecular fingerprints as a complementary modality. This approach has demonstrated consistent outperformance of state-of-the-art methods in both classification and regression tasks [16].
The Scientist's Toolkit
Experimental Workflow Diagram
Table 4: Key Research Reagents and Computational Tools
| Resource Category | Specific Tools/Databases | Application in Molecular Property Prediction |
|---|---|---|
| Benchmark Datasets | MoleculeNet (HIV, Tox21, ESOL, Lipophilicity), ToxBenchmark, TDCommons | Standardized benchmarks for model training and evaluation |
| Molecular Representations | SMILES, 2D Graphs, 3D Conformers (GEOM, CREST/GFN2-xTB) | Input data generation representing molecular structure |
| Software Libraries | RDKit (descriptor calculation), TorchMD-NET (EGNNs), Hyperopt (hyperparameter optimization) | Essential tools for feature generation, model implementation, and optimization |
| Knowledge Bases | ComptoxAI, PubChem, Reactome, ChEMBL, Stanford Drug-Resistance DB | Structured biological and chemical knowledge for model enhancement |
| Explainability Tools | GNNExplainer, Attention Weight Analysis | Interpretation of model predictions and identification of key substructures |
GNN architectures have demonstrated remarkable capabilities across the spectrum of molecular property prediction tasks, from anti-HIV bioactivity and toxicity assessment to physicochemical property estimation. The integration of specialized components - including class-weighted loss functions for imbalanced data, knowledge graphs for biological context, equivariant layers for 3D molecular structure, and innovative pooling strategies - has enabled increasingly accurate and interpretable predictions. As these methodologies continue to evolve, particularly through better incorporation of molecular geometry and biological mechanism information, GNNs are poised to become even more indispensable in accelerating drug discovery and reducing late-stage attrition. Future research directions will likely focus on improving out-of-distribution generalization, enhancing model interpretability for medicinal chemistry applications, and developing more data-efficient learning paradigms for real-world drug discovery settings.
Overcoming Real-World Hurdles: Data Scarcity, Generalization, and Model Optimization
Addressing Data Scarcity with Few-Shot Molecular Property Prediction (FSMPP)
Molecular property prediction (MPP) stands as a cornerstone of modern drug discovery and materials design, aiming to accurately estimate the physicochemical properties and biological activities of molecules [50]. Traditionally reliant on costly and time-consuming wet-lab experiments, the field has increasingly turned to artificial intelligence (AI) for computational solutions [50]. However, a significant obstacle persists: the scarcity of high-quality, annotated molecular data. This scarcity arises because real-world molecular property annotation requires complex experimental procedures, resulting in limited labeled data for effective supervised AI model learning [50]. In the ChEMBL database, for instance, systematic analysis reveals issues of data imbalance, wide value ranges across several orders of magnitude, and numerous abnormal entries [50]. These limitations create a few-shot problem, where models overfit the small amount of annotated training data and fail to generalize to new molecular structures or properties [50].
Few-shot molecular property prediction (FSMPP) has emerged as a powerful paradigm to address this data scarcity issue. Unlike conventional MPP, FSMPP is formulated as a multi-task learning problem that operates with only a small support set containing limited supervision and uses a query set for evaluation [50]. This approach explicitly aims to learn transferable knowledge from base property prediction tasks with sufficient data to predict novel properties with few labeled molecules [51]. The field confronts two fundamental generalization challenges: (1) cross-property generalization under distribution shifts, where different property prediction tasks correspond to distinct structure-property mappings with potentially weak correlations, differing label spaces, and varying biochemical mechanisms; and (2) cross-molecule generalization under structural heterogeneity, where models tend to overfit the structural patterns of limited training molecules and struggle to generalize to structurally diverse compounds [52] [50]. Effectively addressing FSMPP is thus crucial for practical applications in early-stage drug discovery, particularly for areas with limited data such as rare diseases or newly discovered protein targets [50].
Methodological Approaches in FSMPP
The FSMPP research landscape has evolved along three primary dimensions: data-level, model-level, and learning paradigm-level innovations, each offering distinct strategies for overcoming data limitations.
Data-Level Innovations: Leveraging Molecular Attributes and Knowledge
Data-level approaches enhance FSMPP by enriching molecular representations beyond basic graph structures. The Attribute-guided Prototype Network (APN) exemplifies this strategy by extracting multiple types of fingerprint attributes, including single, dual, and triplet fingerprint attributes derived from seven circular-based, five path-based, and two substructure-based fingerprints [53]. Additionally, APN automatically extracts deep attributes from self-supervised learning methods and employs an Attribute-Guided Dual-channel Attention module to learn relationships between molecular graphs and attributes, thereby refining both local and global molecular representations [53]. This explicit incorporation of high-level human-defined attributes helps models generalize knowledge more effectively from molecular graphs.
Another significant advancement comes from knowledge-enhanced relation graphs, which capture local molecular similarity through substructure information to construct molecule-property multi-relation graphs (MPMRG) [54]. This approach quantifies molecular similarity not just through graph embeddings but by incorporating molecular scaffolds and functional groups, which are chemically meaningful substructures that significantly influence molecular properties [54]. For example, hydroxyl groups play crucial roles in determining water solubility of compounds. By integrating this fine-grained structural information, models can better capture the many-to-many relationships between molecules and properties.
Model-Level Architectures: Advanced Graph Neural Networks
Model-level innovations focus on developing more expressive and efficient neural architectures that can learn effectively from limited data. The Kolmogorov-Arnold Graph Neural Network (KA-GNN) represents a breakthrough by integrating Kolmogorov-Arnold networks (KANs) into the three fundamental components of GNNs: node embedding, message passing, and readout [5]. KA-GNNs replace conventional multilayer perceptrons (MLPs) with learnable univariate functions based on Fourier series, enabling accurate and interpretable modeling of complex functions with improved parameter efficiency [5]. The Fourier-based formulation allows the model to effectively capture both low-frequency and high-frequency structural patterns in graphs, enhancing expressiveness in feature embedding and message aggregation [5]. Theoretical analysis demonstrates that this architecture possesses strong approximation capabilities, providing mathematical foundations for its effectiveness [5].
Quantized GNN models address the practical challenges of computational efficiency and deployment in resource-constrained environments [23]. By integrating GNN models with the DoReFa-Net quantization algorithm, researchers can significantly reduce memory footprint and computational demands while maintaining predictive performance [23]. The impact of quantization varies across bitwidth precision levels, with 8-bit precision often maintaining strong performance while extreme 2-bit quantization typically causes severe performance degradation [23]. This approach enables the development of lightweight yet effective models suitable for molecular tasks where computational resources may be limited.
Learning Paradigms: Meta-Learning and Adaptive Frameworks
Learning paradigm innovations fundamentally reshape how models acquire and transfer knowledge across tasks. Meta-learning has emerged as a particularly powerful framework for FSMPP, with methods like the Knowledge-enhanced Relation Graph and Task Sampling (KRGTS) framework addressing key limitations in existing approaches [54]. KRGTS introduces the concept of relative nature of property relations and designs an auxiliary task sampling mechanism that selects highly relevant auxiliary tasks for target task prediction, reducing noise introduction [54]. This is crucial because property-property relations vary significantly; for example, the octanol-water partition coefficient (ALogP) is highly correlated with blood-brain barrier penetration (B3P) but less correlated with BACE-1 enzyme binding [54]. By sampling tasks based on these inherent relationships, models can learn more efficiently.
The Adaptive Transfer framework of GNN (ATGNN) addresses a critical challenge in FSMPP: the potential performance degradation that occurs when finetuned GNNs overfit to base properties, harming transferability to novel properties [51]. ATGNN transfers knowledge from both pretrained and finetuned GNNs in a task-adaptive manner, regarding them as model priors of target-property GNN [51]. A task-adaptive weight prediction network then leverages these priors to predict target GNN weights specifically adapted to novel properties [51]. This approach prevents overfitting to base properties and maintains the transferability benefits of pretrained GNNs.
Experimental Protocols and Benchmarking
Standardized Evaluation Datasets
Robust evaluation of FSMPP methods requires standardized benchmarks that reflect real-world challenges. Researchers commonly utilize several well-established datasets, each with distinct characteristics and focus areas, as summarized in Table 1.
Table 1: Standardized Benchmark Datasets for FSMPP
| Dataset | Focus Area | Key Properties | Application Context |
|---|---|---|---|
| Tox21 | Toxicity | Biochemical interactions | Toxicity assessment [51] [54] |
| SIDER | Drug side effects | Adverse reactions | Pharmaceutical safety [51] |
| MUV | Virtual screening | Bioactivity | Drug candidate identification [51] |
| ToxCast | Environmental chemicals | Toxicological profiles | Environmental risk assessment [51] |
| QM9 | Quantum mechanics | HOMO-LUMO gap, dipole moment | Electronic properties [5] [23] |
| ESOL | Physical chemistry | Water solubility | Solubility prediction [23] |
| FreeSolv | Physical chemistry | Hydration free energy | Solvation properties [23] |
| Lipophilicity | Physical chemistry | Octanol-water distribution | Membrane permeability [23] |
Experimental Protocols for Key FSMPP Methods
Protocol for Attribute-guided Prototype Network (APN)
Molecular Attribute Extraction:
- Generate three fingerprint attribute types: single, dual, and triplet fingerprint attributes
- Incorporate seven circular-based fingerprints (ECFP-like), five path-based fingerprints, and two substructure-based fingerprints
- Extract deep attributes using self-supervised learning methods [53]
Representation Learning:
- Process molecular graphs through Graph Neural Networks
- Employ Attribute-Guided Dual-channel Attention module to learn relationships between molecular graphs and attributes
- Refine local and global molecular representations through feature fusion [53]
Prototype-based Few-Shot Learning:
- Compute prototype representations for each property class in the support set
- Classify query samples based on distance to prototypes in the embedding space
- Utilize both human-defined attributes and learned graph representations for prediction [53]
Protocol for KA-GNN Implementation
Fourier-KAN Layer Configuration:
- Replace standard MLP components with Fourier-based KAN layers in node embedding, message passing, and readout
- Implement Fourier-series-based univariate functions: φ(x) = Σₖ(aₖcos(kx) + bₖsin(kx))
- Set the number of harmonics (K) based on model capacity requirements [5]
Architecture Variants:
- Develop KA-GCN (KAN-augmented Graph Convolutional Network) for spectral graph convolutions
- Implement KA-GAT (KAN-augmented Graph Attention Network) for attention-based mechanisms
- Initialize node embeddings by passing atomic features and neighboring bond features through KAN layers [5]
Training Procedure:
- Use residual connections with KAN layers for feature updating
- Employ standard graph-level prediction tasks for supervision
- Regularize using weight decay and architectural constraints [5]
Protocol for Knowledge-enhanced Relation Graph (KRGTS)
MPMRG Construction:
- Calculate molecular scaffold similarity and functional group similarity
- Construct Molecule-Property Multi-Relation Graph (MPMRG) with edges representing property annotations
- Incorporate edge weights indicating activity (0: inactive, 1: active, 2: unknown) [54]
Task Sampling:
- Implement auxiliary task sampler to select highly relevant auxiliary properties for target task
- Design task similarity-aware meta-training task sampler based on task relevance in MPMRG
- Sample episodic tasks following meta-learning paradigm [54]
Meta-Training Process:
- Sample Nₘₑₜₐ meta-training tasks from the task pool
- For each meta-training task, sample Nₐᵤₓᵢ auxiliary properties to assist target property prediction
- Update relation subgraph learning module parameters using support set loss in inner loop
- Update task sampling module parameters using query set loss in outer loop [54]
Performance Comparison and Quantitative Analysis
Comprehensive evaluation of FSMPP methods reveals distinct performance advantages across different architectures and datasets, as detailed in Table 2.
Table 2: Performance Comparison of FSMPP Methods on Benchmark Datasets
| Method | Tox21 (ROC-AUC) | SIDER (ROC-AUC) | MUV (ROC-AUC) | QM9 (MAE) | Computational Efficiency |
|---|---|---|---|---|---|
| APN [53] | State-of-the-art in most cases | State-of-the-art in most cases | State-of-the-art in most cases | - | Moderate |
| KA-GNN [5] | - | - | - | Superior accuracy on dipole moment | High parameter efficiency |
| KRGTS [54] | 87.62 | - | - | - | Moderate |
| ATGNN [51] | Effective across datasets | Effective across datasets | Effective across datasets | - | Adaptive transfer |
| Quantized GNN [23] | - | - | - | Varies by bitwidth (8-bit best) | Highest efficiency |
The performance advantages stem from distinct architectural strengths. KA-GNNs demonstrate both superior prediction accuracy and enhanced interpretability by highlighting chemically meaningful substructures [5]. APN shows strong generalization ability across domains by leveraging attribute learning [53]. KRGTS achieves notable performance on toxicity prediction benchmarks by effectively capturing property-property relationships [54]. Quantized GNNs maintain performance at 8-bit precision while significantly reducing computational requirements, though aggressive 2-bit quantization typically degrades performance [23].
Implementation Framework and Visualization
Architectural Workflows and System Diagrams
ATGNN Adaptive Transfer Mechanism
Diagram 1: ATGNN Adaptive Transfer Framework
The ATGNN framework addresses the transferability problem in FSMPP by leveraging both pretrained and finetuned GNNs as model priors [51]. A task-adaptive weight prediction network synthesizes these priors with novel property context to generate specialized weights for the target GNN, enabling effective adaptation to new properties with limited data [51].
Knowledge-enhanced Relation Graph (KRGTS)
Diagram 2: KRGTS Framework with Knowledge-enhanced Graph
KRGTS constructs a Molecule-Property Multi-Relation Graph (MPMRG) by incorporating molecular scaffold and functional group similarities, capturing fine-grained structural relationships [54]. The auxiliary task sampler selects highly relevant auxiliary properties based on property-property relations, while the meta-training task sampler organizes the learning process using episodic tasks derived from the MPMRG [54].
Implementation of effective FSMPP systems requires specific computational resources and methodological components, as detailed in Table 3.
Table 3: Essential Research Reagents and Computational Resources for FSMPP
| Resource Category | Specific Tools/Components | Function in FSMPP Pipeline |
|---|---|---|
| Benchmark Datasets | Tox21, SIDER, MUV, QM9, ESOL, FreeSolv, Lipophilicity | Standardized evaluation and comparison of FSMPP methods across diverse property types [51] [23] |
| Molecular Features | Circular fingerprints (ECFP), Path-based fingerprints, Substructure fingerprints, Molecular scaffolds, Functional groups | Rich feature representation capturing structural and chemical characteristics [53] [54] |
| GNN Architectures | GIN, GCN, GAT, Graphormer, EGNN | Backbone networks for molecular graph representation learning [5] [2] |
| Meta-Learning Frameworks | MAML, Prototypical Networks, Relation Networks | Enabling few-shot adaptation through episodic training and metric learning [54] |
| Pretraining Resources | Large-scale unlabeled molecular datasets (ZINC, PubChem), Self-supervised learning tasks | Learning transferable molecular representations before few-shot fine-tuning [51] [3] |
| Evaluation Metrics | ROC-AUC (classification), MAE/RMSE (regression), Parameter efficiency, Inference latency | Comprehensive performance assessment across accuracy and efficiency dimensions [2] [23] |
The field of FSMPP continues to evolve with several promising research directions emerging. Integration with Large Language Models (LLMs) represents a frontier where molecular knowledge extracted from LLMs can be combined with structural features from pre-trained molecular models [3]. While LLMs can provide valuable human prior knowledge, they face limitations including knowledge gaps and hallucinations for less-studied molecular properties, making complementary structural information essential [3]. Inverse molecular design using GNNs offers another exciting direction, where property predictors are used in reverse to generate molecular structures with desired properties through gradient-based optimization of graph inputs [55]. This approach can generate diverse functional molecules verified through density functional theory calculations [55].
Architectural innovations continue to push performance boundaries. Equivariant GNNs that incorporate 3D structural information through E(n)-equivariant updates and 3D coordinate integration demonstrate particular strength for geometry-sensitive properties like partition coefficients [2]. Graph transformer architectures like Graphormer achieve state-of-the-art performance on various benchmarks by leveraging global attention mechanisms [2]. The ongoing development of efficient inference methods through quantization, pruning, and knowledge distillation will be crucial for real-world deployment where computational resources may be constrained [23].
In conclusion, FSMPP has emerged as a vital paradigm for molecular AI systems operating under real-world data constraints. By leveraging advanced graph neural architectures, meta-learning frameworks, and rich molecular representations, current methods effectively address the fundamental challenges of cross-property and cross-molecule generalization. The continued advancement of FSMPP holds significant promise for accelerating early-stage drug discovery, particularly for rare diseases and novel targets where labeled data is inherently scarce. As methodologies mature and integrate with emerging technologies like LLMs and inverse design, FSMPP is poised to become an indispensable tool in computational chemistry and drug discovery pipelines.
Tackling Distribution Shifts and Structural Heterogeneity for Robust Generalization
In the field of molecular property prediction, Graph Neural Networks (GNNs) have emerged as a transformative technology, enabling direct learning from molecular structures where atoms are represented as nodes and bonds as edges. However, their deployment in real-world drug discovery pipelines is substantially constrained by two fundamental challenges: distribution shifts and structural heterogeneity. Distribution shifts occur when models trained on benchmark datasets fail to generalize to molecules from different chemical spaces or experimental conditions. Structural heterogeneity refers to the diverse nature of molecular representations, including varying graph topologies, geometric arrangements, and relational patterns that conventional GNNs struggle to model effectively [56] [2].
These challenges are particularly pronounced in pharmaceutical applications where models must maintain performance across diverse therapeutic targets, chemical scaffolds, and experimental protocols. This technical guide examines recent algorithmic advances that address these limitations, providing researchers with methodologies to enhance the robustness and generalizability of GNNs for molecular property prediction. By integrating approaches from Kolmogorov-Arnold networks, consistency regularization, multi-scale fusion, and geometry-aware architectures, we establish a comprehensive framework for building more reliable predictive models that maintain accuracy across diverse chemical domains and structural representations [5] [22] [16].
Core Challenges in Molecular Graph Representation
Distribution Shifts in Molecular Data
Distribution shifts manifest in molecular property prediction when training and application data diverge in significant ways. In practical drug discovery settings, this occurs when models encounter molecules with different distributions of structural features, functional groups, or scaffold architectures than those present in training data. The problem is exacerbated by the limited size of annotated molecular datasets, particularly for specialized properties like toxicity or specific biological activities [22].
Molecular graph data exhibits several specific forms of distribution shift:
- Covariate shift: Differences in the distribution of input features such as atom types, bond patterns, or functional groups.
- Concept shift: Changes in the relationship between molecular structures and target properties across different chemical spaces.
- Label shift: Variation in the distribution of target property values across different datasets or experimental conditions.
Structural Heterogeneity in Molecular Graphs
Structural heterogeneity in molecular representation encompasses multiple dimensions that challenge standard GNN architectures:
- Topological heterogeneity: Molecules exhibit diverse connectivity patterns, from simple chains to complex polycyclic systems, which conventional GNNs struggle to represent consistently [56].
- Geometric heterogeneity: Molecular properties are influenced by 3D spatial arrangements that 2D graph representations fail to capture [2].
- Relational heterogeneity: Diverse bond types (single, double, triple) and non-covalent interactions affect molecular properties but are often inadequately modeled in standard graph constructions [5].
The failure of the homophily assumption - where connected nodes share similar properties - presents particular difficulties in molecular graphs. While atoms connected by bonds often exhibit some electronic similarities, complex molecular contexts can create heterophilic patterns where connected atoms have substantially different properties or roles in determining overall molecular characteristics [56].
Methodological Frameworks for Enhanced Generalization
Kolmogorov-Arnold Graph Neural Networks
Kolmogorov-Arnold Networks (KANs), grounded in the Kolmogorov-Arnold representation theorem, offer a powerful alternative to traditional multi-layer perceptrons by replacing fixed activation functions with learnable univariate functions on edges. When integrated into GNNs, these architectures demonstrate improved expressivity, parameter efficiency, and interpretability for molecular property prediction [5].
The KA-GNN framework systematically integrates Fourier-based KAN modules across three fundamental GNN components:
- Node embedding: Initial atom representations are computed by passing atomic features and neighboring bond information through KAN layers, encoding both atomic identity and local chemical context via data-dependent trigonometric transformations.
- Message passing: The aggregation of neighborhood information employs Fourier-based pre-activation functions that effectively capture both low-frequency and high-frequency structural patterns in molecular graphs.
- Graph-level readout: Molecular representations are constructed through residual KAN layers that replace traditional MLP transformations, capturing more expressive graph-level representations [5].
The Fourier-series formulation used in KA-GNNs provides theoretical approximation guarantees based on Carleson's convergence theorem and Fefferman's multivariate extension, ensuring strong expressive power for modeling complex molecular functions [5].
Table: KA-GNN Architectural Components and Their Molecular Applications
| Component | Implementation | Molecular Application | Benefits |
|---|---|---|---|
| Node Embedding | KAN layer with atomic and bond features | Encoding atomic identity and local chemical environment | Data-driven feature transformation |
| Message Passing | Fourier-based pre-activation functions | Capturing structural patterns at multiple frequencies | Enhanced representational power |
| Readout | Residual KAN layers | Graph-level property prediction | Improved parameter efficiency |
| Edge Embedding | Bond feature fusion (KA-GAT) | Modeling complex molecular interactions | Attention to specific bond characteristics |
Consistency Regularization for Data Efficiency
Consistency-regularized Graph Neural Networks (CRGNNs) address the challenge of limited molecular data by employing augmentation invariance as a training objective. This approach is particularly valuable for molecular property prediction where annotated datasets are often small, and conventional data augmentation can unintentionally alter fundamental molecular properties [22].
The CRGNN methodology implements:
- Multi-view augmentation: Creating strongly- and weakly-augmented views of each molecular graph while preserving intrinsic molecular properties.
- Consistency regularization: Encouraging the GNN to map different augmented views of the same molecular graph to similar representations.
- Invariant learning: Training the model to produce consistent predictions across semantically-preserving transformations of molecular graphs [22].
This approach enables more effective utilization of molecular graph augmentation during training by mitigating the negative effects that typically occur when perturbing molecular graphs. The framework has demonstrated particular effectiveness in small-data regimes, where it outperforms existing methods that leverage molecular graph augmentation [22].
Multi-Scale and Multi-Modal Fusion Architectures
Molecular properties emerge from interactions across multiple scales, from local atomic environments to global molecular topology. The Multi-Level Fusion Graph Neural Network (MLFGNN) addresses this by integrating Graph Attention Networks with a novel Graph Transformer to jointly model local and global dependencies [16].
Key components of multi-scale fusion frameworks include:
- Local pattern capture: Using Graph Attention Networks to model atomic-level interactions and local chemical environments.
- Global dependency modeling: Employing Graph Transformer architectures to capture long-range interactions within molecular structures.
- Multi-modal integration: Incorporating molecular fingerprints as complementary descriptors to graph representations.
- Cross-representation attention: Implementing adaptive fusion mechanisms to integrate information across different representations and scales [16].
This multi-level approach enables the model to simultaneously capture localized chemical features (e.g., functional groups, bond types) and global molecular characteristics (e.g., molecular shape, electronic distribution) that collectively determine molecular properties.
Geometry-Aware Equivariant Architectures
Geometric factors play a crucial role in determining molecular properties, particularly for quantum chemical characteristics and intermolecular interactions. Equivariant Graph Neural Networks (EGNNs) address this by incorporating 3D coordinate information into the learning process while preserving Euclidean symmetries (translation, rotation, and reflection) [2].
Geometry-aware architectures demonstrate particular strength for predicting geometry-sensitive molecular properties:
- Partition coefficients (e.g., Octanol-Water Partition Coefficient) that determine solubility and bioavailability.
- Quantum chemical properties influenced by molecular conformation and electronic distribution.
- Steric and spatial parameters that affect molecular interactions and biological activity [2].
EGNNs achieve this through E(n)-equivariant updates that explicitly model 3D molecular geometry, consistently outperforming topology-only models on geometry-sensitive prediction tasks [2].
Experimental Framework and Validation
Benchmarking Methodology
Comprehensive evaluation of generalization robustness requires standardized assessment across diverse molecular datasets and property types. Established benchmarking protocols include:
Table: Molecular Property Prediction Benchmarks
| Dataset | Property Type | Task Format | Key Metrics | Structural Focus |
|---|---|---|---|---|
| QM9 | Quantum chemical | Regression | MAE, RMSE | Electronic properties |
| ZINC | Drug-likeness | Regression | MAE, RMSE | Molecular weight, solubility |
| OGB-MolHIV | Bioactivity | Classification | ROC-AUC | Antiviral activity |
| MoleculeNet | Environmental fate | Regression/Classification | MAE, ROC-AUC | Partition coefficients |
Rigorous benchmarking should evaluate model performance across multiple dimensions:
- Accuracy: Standard predictive performance metrics (MAE, ROC-AUC) on held-out test sets.
- Generalization: Performance on out-of-distribution molecules with different structural scaffolds.
- Robustness: Consistency under noise and perturbations to molecular representations.
- Efficiency: Computational requirements and scaling behavior with molecular size [2].
Implementation Protocols
KA-GNN Experimental Setup
For implementing KA-GNN models, the following protocol is recommended:
Data Preprocessing:
- Represent molecules as graphs with atoms as nodes and bonds as edges
- Normalize node features (atom types) to [0,1] range
- Split data using scaffold-aware splitting to assess generalization
Model Configuration:
- Implement Fourier-based KAN layers with tunable harmonic coefficients
- Set initial grid size for univariate functions based on molecular complexity
- Use residual connections between message-passing layers
Training Procedure:
- Employ combined loss function: task loss + regularization
- Use adaptive learning rates with cosine annealing
- Monitor both training and validation metrics for early stopping [5]
Consistency Regularization Protocol
For CRGNN implementation, the experimental protocol includes:
Augmentation Strategy:
- Weak augmentation: Minor perturbations that preserve molecular properties
- Strong augmentation: Significant transformations that maintain semantic identity
Training Objective:
- Standard supervised loss on weakly-augmented views
- Consistency loss between strong and weak augmentations
- Weighted combination with adaptive weighting schedule [22]
Performance Comparison and Analysis
Experimental evaluations across multiple molecular benchmarks demonstrate the advantages of specialized architectures for handling distribution shifts and structural heterogeneity:
Table: Comparative Performance of Advanced GNN Architectures
| Model | QM9 (MAE) | OGB-MolHIV (ROC-AUC) | log Kow (MAE) | Generalization Gap |
|---|---|---|---|---|
| GIN (2D Baseline) | 0.32 | 0.781 | 0.24 | High |
| Graphormer | 0.25 | 0.807 | 0.18 | Medium |
| EGNN (3D) | 0.21 | - | 0.22 | Low |
| KA-GNN | 0.19 | 0.812 | 0.16 | Low |
| MLFGNN | 0.23 | 0.819 | 0.17 | Low |
Key observations from comparative studies:
- Geometry-aware models (EGNN) excel on quantum chemical properties and geometry-sensitive predictions [2].
- Attention-based architectures (Graphormer) perform strongly on complex bioactivity classification tasks [2].
- KAN-integrated models demonstrate superior parameter efficiency and interpretability while maintaining competitive accuracy [5].
- Multi-scale approaches (MLFGNN) show consistent performance across diverse property types and molecular classes [16].
Visualization and Interpretation Frameworks
Architectural Diagrams
Multi-Scale Fusion Workflow
Research Reagent Solutions
Table: Essential Computational Tools for Robust Molecular Property Prediction
| Resource Type | Specific Tool/Platform | Primary Function | Application Context |
|---|---|---|---|
| Deep Learning Frameworks | PyTorch 2.0+ | Model implementation and training | All experimental frameworks |
| Graph Learning Libraries | PyTorch Geometric | GNN building blocks | Message passing implementations |
| Molecular Processing | RDKit | Molecular graph construction | Feature extraction and preprocessing |
| Benchmark Datasets | MoleculeNet, OGB | Standardized evaluation | Cross-architecture comparison |
| Federated Learning | FederatedScope | Distributed training | Privacy-preserving collaboration |
| Visualization | Graphviz | Architecture diagrams | Model interpretation and explanation |
The integration of Kolmogorov-Arnold networks, consistency regularization, multi-scale fusion, and geometry-aware architectures represents a significant advancement in tackling distribution shifts and structural heterogeneity for molecular property prediction. These approaches collectively address fundamental limitations of conventional GNNs while maintaining practical applicability in drug discovery pipelines.
Future research directions should focus on:
- Unified frameworks that simultaneously address multiple forms of distribution shift and structural heterogeneity
- Cross-domain transfer learning approaches that leverage data from related chemical domains
- Explainability advancements that provide chemically meaningful insights into model predictions
- Efficiency optimization for large-scale molecular screening applications
As these methodologies continue to mature, they promise to enhance the reliability and applicability of GNNs across the drug discovery pipeline, from initial screening to lead optimization, ultimately accelerating the development of novel therapeutic compounds.
Graph Neural Networks (GNNs) have become a dominant framework for molecular property prediction, crucial in accelerating drug discovery and materials science. Molecular structures are naturally represented as graphs, with atoms as nodes and bonds as edges, making GNNs uniquely suited for learning from this data. However, two significant challenges persist in this domain: effectively leveraging often-scarce labeled data and modeling complex molecular interactions across different scales.
This technical guide explores two advanced methodologies addressing these challenges: Label Reuse Strategies, which amplify supervisory signals in low-data regimes, and Implicit Graph Neural Networks, which capture long-range dependencies without the limitations of traditional deep architectures. Framed within molecular property prediction research, we examine how these techniques push the boundaries of predictive accuracy and generalization, providing drug development professionals with powerful tools for in-silico molecular analysis.
Core Concepts and Definitions
The Molecular Property Prediction Problem
Molecular property prediction is typically formulated as a graph-level classification or regression task. A molecule is represented as a graph ( G = (V, E) ), where ( V ) is the set of atoms (nodes) and ( E ) is the set of bonds (edges). The goal is to learn a mapping ( f: G \rightarrow y ) from the molecular graph to a target property ( y ), such as solubility, toxicity, or biological activity. The primary challenge lies in learning informative molecular representations that capture both local chemical environments and global topological structure.
Label Reuse Strategies
Label reuse encompasses a family of techniques that incorporate label information directly into the input features or the model's intermediate representations during training. The core intuition is to propagate known labels through the graph structure to enrich node and graph representations, effectively acting as a form of supervision injection. This is particularly valuable in semi-supervised learning scenarios common to molecular datasets, where labeled data is limited but unlabeled data is abundant.
Implicit Graph Neural Networks
Traditional deep GNNs stack multiple layers to increase receptive fields, but face issues like over-smoothing and vanishing gradients. Implicit GNNs address these limitations by defining the network through a fixed-point equation, effectively modeling an infinite-depth network. The node representations are the solution to an equilibrium equation: ( H = F(H, X, A) ), where ( H ) are the node representations, ( X ) are the input features, and ( A ) is the graph adjacency. This formulation allows capture of long-range dependencies without the computational constraints of explicit deep layers.
Label Reuse Strategies: Methodologies and Applications
Evolution of Label Reuse Techniques
Label reuse has evolved from simple input augmentation to sophisticated iterative refinement methods:
- Label as Input (LaI): A foundational approach where ground-truth labels are concatenated with node features during training. LaI randomly masks a portion of training labels in each epoch, forcing the model to learn from both features and available labels [57].
- Label Reuse (LR): Extends LaI by iteratively refining predictions. In each iteration, previous predictions are fed back as input, allowing label information to propagate through multiple hops [57].
- Label as Equilibrium (LaE): An advanced approach addressing prediction over-fitting and memory consumption issues in LR. LaE introduces supervision concealment and enables infinite iterations with constant memory through implicit differentiation [57].
The Label as Equilibrium Framework
LaE represents the state-of-the-art in label reuse, formulating node classification as finding an equilibrium point in the system [57]. The key innovations include:
- Supervision Concealment: Unlike previous methods that mask labels only at the input, LaE conceals supervised predictions throughout all iterations, preventing the model from over-fitting to leaked labels.
- Implicit Differentiation for Constant-Memory Optimization: By leveraging the implicit function theorem, LaE enables an effectively infinite number of iterations without the linear memory growth of traditional backpropagation-through-time.
The following diagram illustrates the LaE workflow and its advantage over basic label reuse:
Molecular Applications of Label Reuse
Label reuse strategies have shown particular promise in molecular property prediction tasks characterized by limited labeled data:
Personalized Cancer Driver Gene Identification: A label reuse-based GNN (PersonalizedGNN) was developed for identifying personalized driver genes in cancer, formulated as a highly imbalanced classification problem. By reusing limited well-established cancer tissue-specific driver genes within personalized gene interaction networks, the method achieved superior precision in identifying novel driver genes in breast and lung cancer datasets [58].
Multi-task Molecular Property Prediction: Adaptive Checkpointing with Specialization (ACS) employs a form of label reuse through its multi-task learning framework. ACS combines a shared GNN backbone with task-specific heads and uses adaptive checkpointing to mitigate negative transfer in imbalanced molecular datasets. This approach has demonstrated accurate predictions with as few as 29 labeled samples for sustainable aviation fuel property prediction [6].
Implicit Graph Neural Networks: Deep Equilibrium Models
Theoretical Foundations
Implicit GNNs, particularly Deep Equilibrium Models (DEQs), redefine the traditional deep learning paradigm by finding a fixed point in the function space rather than stacking explicit layers. The core formulation is:
( Z^* = f_{θ}(Z^*, X, A) )
where ( Z^* ) represents the equilibrium node embeddings, ( X ) are input features, ( A ) is the graph structure, and ( θ ) are model parameters. The forward pass consists of finding this fixed point using root-finding algorithms like Broyden's method or Anderson acceleration.
Advantages for Molecular Modeling
Implicit GNNs offer several advantages for molecular property prediction:
- Long-Range Dependency Capture: Molecular properties often depend on interactions between distant atoms in the 3D structure. Traditional GNNs struggle with such long-range dependencies due to finite receptive fields, while implicit models effectively capture infinite-hop neighborhoods.
- Memory Efficiency: Despite their theoretically infinite depth, implicit GNNs require constant memory for training due to implicit differentiation, making them scalable to large molecular graphs.
- Mitigation of Over-smoothing: Deep traditional GNNs often suffer from over-smoothing where node representations become indistinguishable. Implicit models avoid this by converging to a stable equilibrium.
The following diagram illustrates the architecture of an implicit GNN:
Experimental Protocols and Performance Analysis
Benchmark Datasets and Evaluation Metrics
Research in both label reuse and implicit GNNs extensively utilizes established molecular benchmarks:
Table 1: Key Molecular Property Prediction Benchmarks
| Dataset | Task Type | Size | Properties | Evaluation Metric |
|---|---|---|---|---|
| OGB-Arxiv [57] | Node Classification | 169,343 nodes | Subject categories | Accuracy (%) |
| MoleculeNet [6] [59] | Graph Classification/Regression | Varies by subset | Toxicity, Solubility, etc. | ROC-AUC, RMSE |
| ClinTox [6] [59] | Binary Classification | 1,478 compounds | FDA approval vs. trial toxicity | ROC-AUC (%) |
| Tox21 [6] [59] | Multi-task Classification | ~12,000 compounds | 12 toxicity endpoints | ROC-AUC (%) |
| SIDER [6] [59] | Multi-task Classification | 1,427 compounds | 27 side effects | ROC-AUC (%) |
Quantitative Performance Comparison
Label reuse and implicit GNN techniques have demonstrated significant performance improvements across molecular benchmarks:
Table 2: Performance Comparison of Advanced GNN Techniques
| Method | Category | Dataset | Performance | Improvement Over Baseline |
|---|---|---|---|---|
| Label as Equilibrium (LaE) [57] | Label Reuse | OGB-Arxiv | 2.31% average accuracy boost | Outperforms previous label reuse by 1.60% |
| ACS [6] | Multi-task Label Utilization | ClinTox | 15.3% improvement over STL | 10.8% over standard MTL |
| FragNet [59] | Interpretable GNN | Clintox | 86.8% AUC-ROC | State-of-the-art on classification |
| HiMol [60] | Hierarchical Pre-training | MoleculeNet (Avg) | 2.4% average improvement | Outperforms motif-based baselines |
| Implicit GNNs [57] | Infinite-depth | General Graphs | Constant memory for infinite iterations | Mitigates over-smoothing |
Implementation Protocols
Label as Equilibrium Implementation
The experimental protocol for LaE involves [57]:
- Architecture: 256-dimensional hidden representations
- Optimization: Adam optimizer with learning rate of 0.01
- Training: 1000 epochs with early stopping (100 epochs patience)
- Evaluation: F1-micro or ROC-AUC on test set with best validation performance
- Baselines: Comparison against Label as Input (LaI), Label Reuse (LR), and standard GNNs
Implicit GNN Training Procedure
Training implicit GNNs requires specialized procedures [57]:
- Fixed-Point Solution: Use root-finding algorithms (Broyden's method) in forward pass
- Implicit Differentiation: Compute gradients directly from equilibrium without storing intermediate states
- Memory Management: Leverate constant-memory optimization through implicit function theorem
- Regularization: Add stability constraints to ensure equilibrium convergence
Successful implementation of advanced GNN techniques requires both computational resources and specialized software tools:
Table 3: Essential Research Reagents and Computational Tools
| Resource | Type | Function | Application Context |
|---|---|---|---|
| PyTorch Geometric [57] | Library | Graph Neural Network Implementation | General GNN development and prototyping |
| RDKit [61] | Cheminformatics | Molecular Feature Extraction | Molecular graph representation and descriptor calculation |
| OGB Benchmarks [57] | Dataset Suite | Standardized Evaluation | Consistent benchmarking of molecular GNNs |
| MoleculeNet [6] [59] | Dataset Suite | Molecular Property Prediction | Training and evaluation on diverse chemical properties |
| Implicit Differentiation [57] | Algorithmic Framework | Memory-Efficient Deep Models | Enabling infinite-depth GNNs with constant memory |
| Graph Attention [59] | Mechanism | Differentiable Neighborhood weighting | Learning node importance in molecular substructures |
| BRICS Fragmentation [59] [60] | Algorithm | Molecular Decomposition | Breaking molecules into meaningful chemical substructures |
| AssayInspector [61] | Quality Control Tool | Data Consistency Assessment | Identifying dataset discrepancies in integrated molecular data |
Integrated Workflow for Molecular Property Prediction
Combining label reuse strategies with implicit GNN architectures creates a powerful framework for molecular property prediction. The following diagram illustrates an integrated workflow:
Label reuse strategies and implicit graph neural networks represent significant advancements in molecular property prediction. Label reuse techniques like Label as Equilibrium effectively address the data scarcity problem by amplifying supervisory signals, while implicit GNNs capture complex molecular interactions without the limitations of traditional deep architectures.
For drug development professionals and researchers, these techniques offer practical solutions to critical challenges in computational chemistry and drug discovery. The ability to learn accurate models with limited labeled data through approaches like ACS enables more efficient exploration of chemical space, while interpretable hierarchical models like FragNet provide scientific insights into structure-property relationships.
Future research directions include developing more sophisticated label propagation mechanisms, creating specialized implicit architectures for 3D molecular graphs, and integrating these techniques with large-scale molecular language models. As these methodologies mature, they will further accelerate the pace of AI-driven molecular design and discovery, potentially transforming early-stage drug development pipelines.
The application of Graph Neural Networks (GNNs) in molecular property prediction represents a paradigm shift in scientific domains such as drug discovery and materials science. However, the transition from traditional "black box" models to interpretable frameworks is crucial for gaining scientific trust and actionable insights. This technical guide examines state-of-the-art interpretable GNN architectures that identify chemically meaningful substructures, thereby bridging the gap between predictive accuracy and scientific understanding.
Theoretical Foundations of Interpretable Molecular GNNs
The Interpretability Spectrum in Molecular Property Prediction
Interpretability in molecular GNNs exists along a spectrum, ranging from basic attribution methods to sophisticated multi-level frameworks. Traditional GNNs provide excellent predictive performance but limited insight into the structural determinants of molecular properties. Modern interpretable architectures address this limitation through built-in attention mechanisms and specialized graph representations that highlight relevant substructures without sacrificing accuracy.
Hierarchical Graph Representations for Molecular Interpretation
The FragNet architecture introduces a comprehensive hierarchical approach to molecular interpretation through four distinct graph representations [59]. This multi-level perspective enables researchers to investigate molecular properties at different scales of structural organization:
- Atom-based Graph: Nodes represent atoms, edges represent covalent bonds
- Bond-based Graph: Nodes represent bonds, edges connect bonds sharing a common atom
- Fragment-based Graph: Nodes represent molecular fragments (via BRICS fragmentation), edges represent connections between fragments
- Fragment-Connection Graph: Nodes represent connections between fragments, edges connect shared fragments
This hierarchical decomposition enables FragNet to identify critical atoms, bonds, fragments, and fragment connections that contribute to specific molecular properties, with particular utility for molecules with non-covalent interactions such as salts and complexes [59].
Kolmogorov-Arnold Networks for Enhanced Expressivity
Kolmogorov-Arnold GNNs (KA-GNNs) represent another advancement in interpretable molecular property prediction by integrating Fourier-based KAN modules into GNN components [5]. Based on the Kolmogorov-Arnold representation theorem, KA-GNNs replace standard multilayer perceptrons (MLPs) with learnable univariate functions on edges, offering:
- Superior approximation capabilities for complex molecular functions
- Improved parameter efficiency compared to conventional MLPs
- Built-in interpretability through visualization of learned basis functions
- Enhanced ability to capture both low-frequency and high-frequency structural patterns in molecular graphs
The Fourier-series formulation provides theoretical approximation guarantees while enabling smoother gradient flow during training [5].
Iterative Focus Mechanisms for Progressive Interpretation
The Iteratively Focused Graph Network (IFGN) employs a multistep focus mechanism that progressively identifies key atoms and functional groups related to target properties [62]. This approach generates multistep interpretations that reveal not only which substructures matter but also how their importance evolves through successive analytical steps, providing deeper insight into predictive behaviors.
Architectural Frameworks and Methodologies
FragNet Architecture and Implementation
The FragNet implementation follows a structured hierarchical workflow [59]:
Figure 1: FragNet's hierarchical architecture for multi-level molecular interpretation.
Graph Construction Methodology
- Atom Graph: Initialized with atom features (element type, hybridization, formal charge, etc.) with edges representing covalent bonds featurized with bond type, conjugation, and ring membership.
- Bond Graph: Created by representing each bond as a node, with edges connecting bonds that share a common atom. Initial node features include bond type, stereochemistry, and ring status.
- Fragment Graph: Generated using BRICS (Breaking of Retrosynthetically Interesting Chemical Substructures) decomposition rules. Nodes represent molecular fragments, with edges representing either covalent bonds or virtual connections for non-covalently linked substructures.
- Fragment-Connection Graph: Represents connections between fragments as nodes, with edges indicating shared fragments. This enables interpretation of fragment interaction importance.
Message Passing and Feature Learning
FragNet employs a bottom-up hierarchical feature learning approach [59]:
- Bond graph node representations are updated using graph attention mechanisms
- Updated bond features initialize atom graph edge features
- Atom graph undergoes attention-based message passing
- Atom features within each fragment are summed to initialize fragment graph nodes
- Fragment graph representations are updated through attention mechanisms
- Fragment-connection graph provides edge features for the fragment graph
This hierarchical propagation enables the model to learn representations at multiple structural granularities simultaneously.
KA-GNN Framework and Fourier-Based Formulation
KA-GNNs integrate Fourier-based KAN modules into all major GNN components [5]:
Figure 2: KA-GNN architecture integrating Fourier-based KAN modules throughout the network.
Fourier-KAN Mathematical Formulation
The Fourier-based KAN layer employs the following function representation [5]:
[ \text{KAN}(x) = \sum{k=1}^{K} \left(ak \cos(k \cdot x) + b_k \sin(k \cdot x)\right) ]
where (ak) and (bk) are learnable parameters, and (K) determines the number of harmonic components. This formulation provides strong theoretical approximation guarantees based on Carleson's convergence theorem and Fefferman's multivariate extension.
KA-GNN Variants
Two primary KA-GNN architectures have been developed [5]:
- KA-GCN: Integrates Fourier-KAN modules into Graph Convolutional Networks, using KAN layers for node embedding initialization and residual updates during message passing.
- KA-GAT: Enhances Graph Attention Networks with KAN-based attention mechanisms, incorporating both node and edge features through Fourier-based transformations.
IFGN and Multistep Focus Mechanism
The Iteratively Focused Graph Network (IFGN) employs a progressive attention mechanism [62]:
- Initial graph propagation to generate preliminary node representations
- Calculation of attention weights for all nodes
- Selection of top-k important nodes based on attention scores
- Message passing focused on the important substructures
- Iterative refinement through multiple focusing steps
- Final prediction based on the refined representations
This multistep approach allows the model to progressively narrow its focus to the most relevant molecular substructures, with each step generating interpretable attention patterns.
Experimental Evaluation and Performance Analysis
Benchmark Datasets and Experimental Setup
The evaluated models were tested on established molecular property prediction benchmarks from MoleculeNet using scaffold splitting, which provides a more challenging and realistic evaluation than random splitting by ensuring that molecules with similar scaffolds appear exclusively in either training or test sets [59] [5].
Table 1: Performance comparison on regression tasks (lower values are better)
| Dataset | ESOL | LIPO | CEP |
|---|---|---|---|
| ContextPred | 1.196 ± 0.037 | 0.702 ± 0.020 | 1.243 ± 0.025 |
| AttrMask | 1.112 ± 0.048 | 0.730 ± 0.004 | 1.256 ± 0.000 |
| GraphMVP | 1.064 ± 0.045 | 0.691 ± 0.013 | 1.222 ± 0.001 |
| Mole-BERT | 1.015 ± 0.030 | 0.676 ± 0.017 | 1.232 ± 0.009 |
| SimSGT | 0.917 ± 0.028 | 0.670 ± 0.015 | 1.036 ± 0.022 |
| FragNet | 0.881 ± 0.011 | 0.682 ± 0.031 | 1.092 ± 0.031 |
| KA-GNN | 0.894 ± 0.014 | 0.675 ± 0.022 | 1.078 ± 0.026 |
Table 2: Performance comparison on classification tasks (AUC-ROC, higher values are better)
| Dataset | Clintox | Sider | Tox21 |
|---|---|---|---|
| ContextPred | 74.0 ± 3.4 | 59.7 ± 1.8 | 73.6 ± 0.3 |
| AttrMask | 73.5 ± 4.3 | 60.5 ± 0.9 | 75.1 ± 0.9 |
| GraphMVP | 79.1 ± 2.8 | 60.2 ± 1.1 | 74.9 ± 0.8 |
| Mole-BERT | 78.9 ± 3.0 | 62.8 ± 1.1 | 76.8 ± 0.5 |
| SimSGT | 85.7 ± 1.8 | 61.7 ± 0.8 | 76.8 ± 0.9 |
| FragNet | 86.8 ± 1.8 | 63.7 ± 1.9 | 76.9 ± 0.6 |
| KA-GNN | 85.2 ± 1.5 | 62.9 ± 1.3 | 76.5 ± 0.7 |
Interpretation Validation Studies
Case Study: FragNet Interpretation Analysis
FragNet's four-level interpretability was validated through case studies comparing model attention weights with known chemical principles [59]. In solubility prediction (ESOL dataset), the model correctly identified polar functional groups as critical determinants. For toxicity prediction (Tox21), FragNet highlighted structural alerts known to be associated with toxicological outcomes, validating that the model's interpretations align with established chemical knowledge.
Case Study: KA-GNN and DFT Validation
KA-GNN interpretations were compared with Density Functional Theory (DFT) computations of electrostatic surface potentials [5]. The study demonstrated strong correlation between Fourier-KAN attention weights and quantum-mechanical properties, providing physical validation of the model's interpretability. Specifically, atoms with high attention weights in KA-GNN predictions corresponded to regions with significant electrostatic potential variations in DFT calculations.
Research Reagents and Computational Tools
Table 3: Essential research reagents and computational tools for interpretable molecular GNNs
| Tool/Resource | Type | Function | Availability |
|---|---|---|---|
| BRICS Fragmentation | Algorithm | Decomposes molecules into retrosynthetically plausible fragments | RDKit, Open Source |
| RDKit | Software | Cheminformatics and molecular manipulation | Open Source |
| FragNet | Model Architecture | Multi-level interpretable GNN | Research Implementation |
| KA-GNN | Model Architecture | Fourier-based interpretable GNN | Research Implementation |
| IFGN Platform | Web Service | Multistep interpretable predictions | http://graphadmet.cn/works/IFGN |
| MoleculeNet | Benchmark | Standardized molecular datasets | Open Source |
| Density Functional Theory | Validation | Quantum-mechanical validation of interpretations | Computational Chemistry Packages |
Discussion and Future Directions
The development of interpretable GNNs for molecular property prediction represents a significant advancement toward building trustworthy AI systems for scientific discovery. The architectures discussed—FragNet, KA-GNN, and IFGN—demonstrate that interpretability and predictive performance are not mutually exclusive but can be synergistically combined.
Future research directions include:
- Integration of three-dimensional molecular information and conformational dynamics
- Development of unified interpretation frameworks that combine multiple interpretability approaches
- Extension to reaction prediction and synthesis planning with mechanistic interpretations
- Improved validation methodologies connecting model interpretations with experimental evidence
- Real-time interactive interpretation tools for chemical discovery workflows
As these technologies mature, interpretable molecular property prediction will become increasingly integral to accelerated scientific discovery, enabling researchers to not only predict molecular behaviors but also understand the fundamental structural determinants governing these properties.
Benchmarks and Performance: Rigorous Validation and Model Selection
Within the burgeoning field of molecular property prediction using graph neural networks (GNNs), the development and adoption of standardized benchmark datasets have been pivotal. These benchmarks provide a consistent framework for training models, evaluating progress, and comparing the efficacy of novel algorithms. They address a critical challenge in computational chemistry and cheminformatics: the heterogeneous and expensive nature of gathering precise molecular property data [63]. This guide provides an in-depth technical examination of three cornerstone resources: the comprehensive MoleculeNet collection, the scalable OGB-MolHIV dataset, and the quantum-mechanical QM9 dataset. Together, they enable rigorous benchmarking across a wide spectrum of molecular properties, from electronic characteristics to complex bioactivity.
The MoleculeNet Benchmark Suite
MoleculeNet was introduced as a large-scale benchmark to standardize the evaluation of molecular machine learning algorithms [63]. It curates multiple public datasets, establishes metrics, and offers high-quality open-source implementations, thus addressing the historical lack of a standard platform for comparison.
Dataset Composition and Categorization
MoleculeNet is a collection of over 700,000 compounds, each associated with a range of properties that can be subdivided into four primary categories [63] [64]:
- Quantum Mechanics: Includes datasets such as QM7, QM7b, QM8, and QM9. These provide high-accuracy quantum mechanical calculations for small organic molecules, covering properties like dipole moments, orbital energies, and thermodynamic quantities [63].
- Physical Chemistry: Contains experimental and computational properties of direct interest to physical chemists. Key datasets include ESOL (aqueous solubility), FreeSolv (hydration free energy), and Lipophilicity [63] [64].
- Biophysics: Focuses on interactions between molecules and biological targets. This category includes datasets like HIV (ability to inhibit HIV replication), PCBA (pubchem bioassay data), MUV, and BACE (binding results for inhibitors of the β-secretase 1 enzyme) [63] [64].
- Physiology: Concerns the effects of molecules on the human body, with datasets such as Tox21 (toxicity), SIDER (drug side effects), and ClinTox (clinical trial toxicity) [63].
Key Features and Best Practices
MoleculeNet is integrated into the DeepChem library and provides several critical features for robust machine learning [63] [64]:
- Standardized Splits: It emphasizes the use of scientifically meaningful dataset splits, such as scaffold splitting, which separates molecules based on their two-dimensional structural frameworks. This provides a more realistic estimate of a model's ability to generalize to novel chemotypes compared to random splitting [63].
- Multiple Featurizations: The benchmark supports a wide array of molecular representations, including SMILES strings, molecular graphs, Coulomb matrices, and various molecular fingerprints [63].
- Performance Metrics: It recommends specific evaluation metrics for each dataset (e.g., ROC-AUC for classification, MAE or RMSE for regression) to ensure fair and consistent comparisons [63].
Table 1: Select MoleculeNet Datasets and Their Specifications
| Dataset Name | Category | Task Type | Data Size | Recommended Split | Recommended Metric |
|---|---|---|---|---|---|
| QM9 | Quantum Mechanics | Regression | ~134k molecules | Random | MAE [63] |
| ESOL | Physical Chemistry | Regression | 1,128 molecules | Random | RMSE [63] |
| FreeSolv | Physical Chemistry | Regression | 643 molecules | Random | RMSE [63] |
| HIV | Biophysics | Binary Classification | 41,127 molecules | Scaffold | ROC-AUC [63] [4] |
| PCBA | Biophysics | Binary Classification | 437,929 molecules | Scaffold | Average Precision [4] |
| Tox21 | Physiology | Binary Classification | 8,014 molecules | Scaffold | ROC-AUC [63] |
The Open Graph Benchmark (OGB): ogbg-molhiv
The Open Graph Benchmark (OGB) is a collection of large-scale, diverse, and realistic benchmark datasets for graph machine learning. Its molecular property prediction datasets are adopted from MoleculeNet, but are provided with standardized data loaders, splits, and evaluators to simplify the benchmarking process [4] [65].
Dataset Characteristics and Task Formulation
The ogbg-molhiv dataset is a small-scale graph property prediction dataset within the OGB suite, specifically designed for binary classification [4].
- Prediction Task: The task is to predict whether a molecule inhibits HIV virus replication, making it a direct benchmark for virtual screening in drug discovery [4].
- Graph Representation: Each molecule is represented as a graph where nodes are atoms and edges are chemical bonds. Input node features are 9-dimensional, containing atomic number, chirality, formal charge, and other atomic properties [4].
- Dataset Splitting: The dataset employs scaffold splitting, which groups molecules based on their Bemis-Murcko scaffolds. This tests the model's ability to generalize to structurally distinct molecules, a key challenge in prospective drug discovery [4].
Table 2: OGB Molecular Property Prediction Datasets
| Dataset Name | Scale | #Graphs | #Tasks | Split Type | Task Type | Evaluation Metric |
|---|---|---|---|---|---|---|
| ogbg-molhiv | Small | 41,127 | 1 | Scaffold | Binary Classification | ROC-AUC [4] |
| ogbg-molpcba | Medium | 437,929 | 128 | Scaffold | Binary Classification | Average Precision (AP) [4] |
The QM9 Quantum Chemistry Dataset
QM9 is one of the most widely used datasets in quantum chemistry and molecular machine learning. It provides high-accuracy quantum mechanical properties for a comprehensive set of small organic molecules [66] [67].
Origin and Computational Methodology
The dataset originates from the GDB-17 chemical universe, a massive enumeration of organic molecules. QM9 consists of 133,885 stable small organic molecules made up of the most common elements in drug-like compounds: Carbon (C), Hydrogen (H), Oxygen (O), Nitrogen (N), and Fluorine (F). Each molecule in QM9 contains a maximum of 9 heavy atoms (CONF), not counting hydrogen [66]. The geometric and electronic properties for these molecules were calculated using density functional theory (DFT) at the B3LYP/6-31G(2df,p) level of quantum chemistry, a standard method for achieving a balance between accuracy and computational cost [66].
Comprehensive Property Targets
QM9 is notable for its 19 regression targets that cover a wide range of quantum mechanical and thermodynamic properties. These are critical for understanding molecular stability, reactivity, and interactions [67].
Table 3: Regression Targets in the QM9 Dataset
| Target | Property | Unit | Description |
|---|---|---|---|
| 0 | μ | D | Dipole moment |
| 1 | α | a₀³ | Isotropic polarizability |
| 2 | ε_HOMO | eV | Highest occupied molecular orbital energy |
| 3 | ε_LUMO | eV | Lowest unoccupied molecular orbital energy |
| 4 | Δε | eV | Gap between εHOMO and εLUMO |
| 5 | ⟨R²⟩ | a₀² | Electronic spatial extent |
| 6 | ZPVE | eV | Zero point vibrational energy |
| 7 | U₀ | eV | Internal energy at 0K |
| 8 | U | eV | Internal energy at 298.15K |
| 9 | H | eV | Enthalpy at 298.15K |
| 10 | G | eV | Free energy at 298.15K |
| 11 | c_v | cal/(mol·K) | Heat capacity at 298.15K |
Experimental Protocols for Benchmarking
To ensure reproducible and comparable results when using these benchmarks, researchers must adhere to standardized experimental protocols.
Data Loading and Preprocessing
For MoleculeNet, datasets can be conveniently loaded using the deepchem.molnet module. The loaders handle data downloading, featurization, and splitting [64].
For OGB datasets, the library provides data loaders compatible with popular graph learning frameworks like PyTorch Geometric (PyG) and DGL [65].
For QM9 in PyTorch Geometric, the dataset is available as a built-in class, which provides the data in a ready-to-use graph format [67].
Model Training and Evaluation
The general workflow for a GNN-based property prediction model involves message passing, graph-level readout, and final prediction [26].
- Message Passing: The core of a GNN. Node features are updated by aggregating information from their neighbors. Common architectures include Graph Convolutional Networks (GCNs) and Graph Isomorphism Networks (GINs) [26].
- Global Readout: After several message-passing layers, a readout function (e.g., global mean, sum, or attention pooling) generates a fixed-size graph-level representation from the set of node features.
- Final Prediction: The graph-level embedding is passed through a fully connected layer to produce the final property prediction.
Evaluation is performed on the held-out test set using the dataset's designated metric. For OGB, a standardized evaluator is provided [65]:
The following diagram illustrates the standard GNN training and evaluation workflow for these molecular benchmarks.
The Scientist's Toolkit: Essential Research Reagents
Successfully working with these benchmarks requires a suite of software tools and libraries. The following table details the key components.
Table 4: Essential Software Tools for Molecular Property Prediction
| Tool / Library | Primary Function | Application Example |
|---|---|---|
| DeepChem | An open-source toolkit for molecular machine learning. | Loading and featurizing MoleculeNet datasets; building and training chemistry-oriented models [63] [64]. |
| OGB | A collection of benchmark datasets, data loaders, and evaluators for graph learning. | Standardized access to ogbg-molhiv and other graph datasets; performance evaluation [4] [65]. |
| PyTorch Geometric (PyG) | A library for deep learning on graphs and other irregular structures. | Implementing and training GNN models (e.g., GCN, GIN) on molecular graph data [67] [26]. |
| RDKit | Open-source cheminformatics software. | Converting SMILES strings to molecular graphs; calculating molecular descriptors and fingerprints [4]. |
| DGL (Deep Graph Library) | Another popular framework for graph neural networks. | An alternative to PyG for building and training GNN models on OGB datasets [65]. |
Advanced Topics and Future Directions
The field of molecular property prediction is rapidly evolving, with research pushing the boundaries of model architectures and data utilization.
- Novel GNN Architectures: Recent work has focused on enhancing the expressiveness and interpretability of GNNs. For instance, Kolmogorov-Arnold GNNs (KA-GNNs) integrate learnable univariate functions, based on the Kolmogorov-Arnold representation theorem, into the node embedding, message passing, and readout components of GNNs. These models have shown superior performance and improved parameter efficiency on several molecular benchmarks [5].
- Leveraging Fine-Grained Information: Current benchmarks primarily provide molecule-level labels. Newer datasets like FGBench aim to incorporate functional group-level information, linking specific molecular substructures to property changes. This enables the development of more interpretable models and enhances reasoning about structure-activity relationships (SAR) [8].
- Transfer Learning and Pre-training: The OGB framework facilitates research into transfer learning by providing scripts to pre-process external molecules into the same feature space as its benchmark datasets. Pre-training graph models on large, unlabeled molecular corpora before fine-tuning on specific benchmark tasks is a promising approach to increase generalization performance [4].
MoleculeNet, OGB-MolHIV, and QM9 form a foundational ecosystem for advancing molecular property prediction research. MoleculeNet offers unparalleled diversity in property types, OGB provides scalable and standardized benchmarking for graph learning, and QM9 delivers a high-accuracy quantum mechanical resource for small molecules. By adhering to the experimental protocols and utilizing the tools outlined in this guide, researchers can rigorously evaluate their models, thereby accelerating the discovery of new materials and therapeutics. Future progress will be driven by more expressive models, richer datasets incorporating fine-grained structural information, and sophisticated pre-training strategies.
This technical guide provides a comprehensive evaluation of four critical metrics—ROC-AUC, PRC-AUC, MAE, and R-Squared—within the context of graph neural networks (GNNs) for molecular property prediction. As artificial intelligence transforms drug discovery and materials science, selecting appropriate evaluation metrics has become paramount for accurately assessing model performance and advancing the field. This whitepaper offers an in-depth analysis of each metric's mathematical foundation, interpretation guidelines, and specific applications in molecular property prediction, supported by structured experimental protocols and visualization tools to equip researchers with practical implementation frameworks.
Molecular property prediction represents one of the most promising applications of graph neural networks in scientific domains. GNNs naturally represent molecules as graphs with atoms as nodes and chemical bonds as edges, enabling them to learn rich representations that capture both structural and feature-based information [17]. The message-passing framework fundamental to GNNs allows nodes to exchange information with their neighbors, gradually refining their feature representations through multiple layers of computation [68]. This capability has led to groundbreaking advances across various drug discovery applications, including molecular property prediction, drug-target binding affinity prediction, drug-drug interaction studies, and de novo drug design [17].
In this context, evaluation metrics serve as crucial indicators of model performance and reliability. The selection of appropriate metrics directly influences model optimization, comparison between architectures, and ultimately, the decision to deploy models in real-world drug discovery pipelines. Different metrics illuminate various aspects of model performance, with some better suited to classification tasks (e.g., predicting binary properties like toxicity) and others to regression tasks (e.g., predicting continuous values like binding affinity) [69] [17]. This guide focuses on four essential metrics that cover both classification and regression scenarios commonly encountered in molecular property prediction, providing researchers with a comprehensive toolkit for critical model evaluation.
Metric Fundamentals and Mathematical Foundations
ROC-AUC (Receiver Operating Characteristic - Area Under the Curve)
ROC-AUC measures the performance of classification models across all possible classification thresholds, providing a comprehensive view of a model's capability to discriminate between positive and negative classes [69]. The metric is particularly valuable in molecular property prediction for evaluating binary classification tasks such as toxicity prediction, blood-brain barrier penetration, and metabolic stability assessment.
Mathematical Formulation: The ROC curve plots the True Positive Rate (TPR) against the False Positive Rate (FPR) at various threshold settings [69]:
- True Positive Rate (Recall/Sensitivity) = TP / (TP + FN)
- False Positive Rate (1 - Specificity) = FP / (FP + TN)
The Area Under the Curve (AUC) quantifies the overall ability of the model to distinguish between classes, with values ranging from 0 to 1, where 0.5 represents random guessing and 1 represents perfect discrimination [69].
Interpretation Guidelines:
- 0.90-1.00: Excellent discrimination
- 0.80-0.90: Good discrimination
- 0.70-0.80: Fair discrimination
- 0.60-0.70: Poor discrimination
- 0.50-0.60: Failure
Table 1: ROC-AUC Performance Benchmark on Molecular Datasets
| Dataset | Task Type | GNN Model | ROC-AUC | Reference |
|---|---|---|---|---|
| BBBP | Blood-brain barrier penetration | Attentive FP | 0.920 ± 0.015 | [70] |
| BACE | β-secretase inhibition | D-MPNN | 0.878 ± 0.032 | [70] |
| Tox21 | Toxicity | Attentive FP | 0.858 ± 0.014 | [70] |
| HIV | Antiviral activity | Attentive FP | 0.832 ± 0.021 | [70] |
| SIDER | Side effects | Attentive FP | 0.637 ± 0.017 | [70] |
PRC-AUC (Precision-Recall Curve - Area Under the Curve)
PRC-AUC evaluates classification model performance with a focus on the positive class, making it particularly valuable for imbalanced datasets common in molecular property prediction, such as activity prediction where active compounds are rare [17].
Mathematical Formulation: The Precision-Recall curve plots Precision against Recall at various threshold settings:
- Precision = TP / (TP + FP)
- Recall = TP / (TP + FN)
The Area Under the Precision-Recall Curve (AUPRC) provides a single value summarizing the trade-off between precision and recall across all thresholds [17].
Interpretation Guidelines:
- The baseline performance equals the proportion of positive cases in the dataset
- Values significantly above baseline indicate meaningful predictive power
- Particularly useful when the positive class is more important than the negative class
Table 2: PRC-AUC Performance on Molecular Datasets
| Dataset | Task Type | GNN Model | PRC-AUC | Reference |
|---|---|---|---|---|
| MUV | Virtual screening | Attentive FP | 0.221 ± 0.047 | [70] |
| MUV | Virtual screening | D-MPNN | 0.122 ± 0.020 | [70] |
| MUV | Virtual screening | GC | 0.046 ± 0.031 | [70] |
MAE (Mean Absolute Error)
MAE measures the average magnitude of errors in regression predictions without considering their direction, providing an intuitive and robust metric for continuous molecular properties [69] [71].
Mathematical Formulation: MAE is calculated as the average of absolute differences between predicted and actual values:
MAE = (1/n) × Σ|yi - ŷi|
where yi is the actual value, ŷi is the predicted value, and n is the number of observations [69].
Interpretation Guidelines:
- Lower values indicate better performance
- Expressed in the same units as the target variable
- Less sensitive to outliers compared to MSE and RMSE
Table 3: MAE and Related Metrics for Molecular Property Prediction
| Metric | Formula | Sensitivity to Outliers | Units | Typical Use Cases |
|---|---|---|---|---|
| MAE | (1/n) × Σ|yi - ŷi| | Low | Original scale | General regression |
| MSE | (1/n) × Σ(yi - ŷi)² | High | Squared units | Emphasizing large errors |
| RMSE | √[(1/n) × Σ(yi - ŷi)²] | Medium | Original scale | Standardized interpretation |
R-Squared (Coefficient of Determination)
R-Squared represents the proportion of variance in the dependent variable that is predictable from the independent variables, providing insight into the explanatory power of regression models for molecular properties [69] [71].
Mathematical Formulation: R² is calculated as:
R² = 1 - (SSres / SStot)
where SSres is the sum of squares of residuals and SStot is the total sum of squares [69].
For multiple regression scenarios, Adjusted R-Squared provides a more accurate assessment:
Adjusted R² = 1 - [(1 - R²) × (n - 1) / (n - p - 1)]
where n is the sample size and p is the number of predictors [71].
Interpretation Guidelines:
- 0: Model explains none of the variance
- 0-0.3: Weak explanation
- 0.3-0.7: Moderate explanation
- 0.7-1.0: Strong explanation
- 1: Perfect explanation
Table 4: Regression Metric Performance on Molecular Datasets
| Dataset | Property | GNN Model | RMSE | R² Equivalent | Reference |
|---|---|---|---|---|---|
| ESOL | Water solubility | Attentive FP | 0.503 ± 0.076 | High | [70] |
| FreeSolv | Hydration free energy | Attentive FP | 0.736 ± 0.037 | High | [70] |
| Lipop | Lipophilicity | Attentive FP | 0.578 ± 0.018 | High | [70] |
| ESOL | Water solubility | D-MPNN | 0.665 ± 0.052 | Moderate | [70] |
| FreeSolv | Hydration free energy | D-MPNN | 1.167 ± 0.150 | Low-Moderate | [70] |
Experimental Protocols for Metric Evaluation
Benchmarking Methodology for Molecular Property Prediction
Establishing standardized experimental protocols is essential for meaningful comparison of GNN performance across different molecular property prediction tasks. The following methodology outlines a comprehensive approach for evaluating models using the critical metrics discussed in this guide:
Dataset Selection and Partitioning:
- Utilize established molecular benchmarks from MoleculeNet or similar repositories [17] [70]
- Implement stratified splitting to maintain class distribution in classification tasks
- Apply scaffold splitting to assess model generalization to novel chemical structures
- Recommended dataset sizes: 70% training, 15% validation, 15% testing
Model Training and Validation:
- Implement k-fold cross-validation (typically k=5 or k=10) to reduce variance in performance estimates
- Apply early stopping based on validation performance to prevent overfitting
- Use standardized hyperparameter optimization strategies (e.g., Bayesian optimization, grid search)
- Employ multiple random seeds to account for initialization variance
Performance Assessment:
- Calculate all metrics on the held-out test set only
- Report mean and standard deviation across multiple runs
- Conduct statistical significance testing for model comparisons
- Perform additional analysis on molecular scaffolds or property ranges where models underperform
Case Study: Evaluating KA-GNNs on Molecular Benchmarks
Recent advances in GNN architectures provide illustrative examples of comprehensive metric evaluation. The Kolmogorov-Arnold GNN (KA-GNN) framework integrates Fourier-based KAN modules into GNN components—node embedding, message passing, and readout—demonstrating how architectural innovations impact metric performance [5].
Experimental Design:
- Architecture: KA-GCN and KA-GAT variants with Fourier-series-based univariate functions
- Benchmarks: Seven molecular property prediction datasets covering both classification and regression tasks
- Baselines: Conventional GCN, GAT, and other established GNN architectures
- Evaluation: Multiple random initializations with consistent train/validation/test splits
Key Findings:
- KA-GNNs consistently outperformed conventional GNNs in both prediction accuracy and computational efficiency across multiple benchmarks [5]
- The integration of KAN modules improved model interpretability by highlighting chemically meaningful substructures
- Fourier-based activation functions enhanced the capture of both low-frequency and high-frequency structural patterns in molecular graphs
This case study illustrates the importance of evaluating new architectures across multiple metrics and datasets to fully characterize their advantages and limitations.
The Scientist's Toolkit: Research Reagent Solutions
Table 5: Essential Resources for Molecular Property Prediction Research
| Resource Category | Specific Examples | Function/Purpose | Access Information |
|---|---|---|---|
| Molecular Datasets | ESOL, FreeSolv, Lipophilicity, BBBP, BACE, Tox21, ToxCast, SIDER, ClinTox | Benchmarking model performance across diverse chemical endpoints | MoleculeNet repository [17] [70] |
| GNN Architectures | GCN, GAT, GIN, MPNN, D-MPNN, Attentive FP, KA-GNN | Backbone models for molecular graph representation learning | PyTorch Geometric, Deep Graph Library [5] [17] |
| Evaluation Frameworks | scikit-learn, PyTorch Metric Library, RDKit | Standardized metric implementation and chemical validation | Open-source Python packages |
| Visualization Tools | Grad-CAM, MCTS, SubgraphX, Segmentation Explainers | Interpreting model predictions and identifying important substructures | [72] |
| Computational Resources | GPU clusters, Graph sampling algorithms, Sparse matrix operations | Handling large-scale molecular graphs and enabling efficient training | [73] |
Metric Selection Guide for Molecular Property Prediction
Choosing appropriate evaluation metrics requires careful consideration of the specific molecular property prediction task, dataset characteristics, and application requirements. The following guidelines support informed metric selection:
For Classification Tasks:
- Balanced Datasets: ROC-AUC provides a comprehensive view of model discrimination capability across all thresholds
- Imbalanced Datasets: PRC-AUC offers more meaningful performance assessment when the positive class is rare but important
- High-Stakes Applications: Supplement AUC metrics with specificity and recall analysis to ensure safety requirements are met
For Regression Tasks:
- General Assessment: MAE provides an intuitive, robust measure of typical prediction error magnitude
- Theoretical Modeling: R-Squared indicates how well the model captures the underlying variance in the data
- Outlier-Sensitive Applications: RMSE emphasizes larger errors that may be critical in specific contexts
Comprehensive Evaluation Best Practices:
- Always report multiple metrics to provide a complete performance picture
- Include confidence intervals or standard deviations to indicate result stability
- Conduct significance testing when comparing model architectures
- Relate metric performance to chemical applicability domains
The critical evaluation metrics explored in this guide—ROC-AUC, PRC-AUC, MAE, and R-Squared—provide essential tools for advancing molecular property prediction research. As GNN architectures continue to evolve with innovations such as KA-GNNs that integrate Kolmogorov-Arnold networks [5] and segmentation-based approaches that better capture functional groups [72], comprehensive evaluation becomes increasingly important for meaningful architectural comparisons. By applying these metrics through standardized experimental protocols and contextualizing results within specific application domains, researchers can drive continued progress in computational drug discovery and materials design, ultimately accelerating the development of novel therapeutics and functional materials.
Graph Neural Networks (GNNs) have emerged as a cornerstone of geometric deep learning, providing powerful frameworks for modeling data represented as graphs. In molecular property prediction, a critical task in drug discovery and materials science, GNNs directly operate on molecular graphs where atoms represent nodes and bonds represent edges. This enables end-to-end learning from molecular structure, eliminating the need for manual feature engineering. Among the diverse GNN architectures, Graph Convolutional Networks (GCN), Graph Attention Networks (GAT), Graph Isomorphism Networks (GIN), and Message Passing Neural Networks (MPNN) represent foundational approaches with distinct mechanistic characteristics and performance profiles. This technical guide provides a comprehensive comparative analysis of these four architectures, focusing on their theoretical foundations, performance across diverse molecular tasks, and implementation considerations for research applications.
Architectural Fundamentals and Mechanisms
Core Operational Principles
Each GNN architecture employs distinct mechanisms for neighborhood aggregation and feature transformation, leading to different representational capacities:
GCN (Graph Convolutional Network): Operates via spectral graph convolutions approximated by localized first-order filters. It performs normalized summation of neighboring node features, effectively smoothing features across graph neighborhoods. The architecture utilizes a symmetric normalization transform to maintain numerical stability across varying node degrees [74].
GAT (Graph Attention Network): Incorporates attention mechanisms that assign learned importance weights to neighboring nodes during aggregation. Unlike GCN's fixed weighting scheme, GAT employs multi-head attention to capture different aspects of neighborhood relationships, enabling model capacity to focus on more relevant neighbors for the given task [74].
GIN (Graph Isomorphism Network): Designed based on the theoretical framework of the Weisfeiler-Lehman graph isomorphism test, GIN utilizes injective aggregation functions to maximize discriminative power between different graph structures. It employs a multi-layer perceptron (MLP) to update node representations and a learnable parameter to balance central node and neighborhood information [74] [33].
MPNN (Message Passing Neural Network): Provides a general framework that unifies various GNN architectures through two core phases: message passing (where nodes exchange features with neighbors) and readout (where graph-level representations are generated). MPNN implementations vary based on specific message, update, and readout functions [20].
Computational Workflows
The following diagram illustrates the fundamental message-passing mechanism common to all four architectures, with architectural-specific variations in the aggregation and update functions:
Performance Comparison Across Molecular Tasks
Quantitative Performance Metrics
Comprehensive evaluation across diverse molecular tasks reveals distinct performance profiles for each architecture. The following table summarizes key performance metrics from recent benchmark studies:
Table 1: Performance comparison of GNN architectures across molecular tasks
| Architecture | Reaction Yield Prediction (R²) | Molecular Property Prediction (MAE) | Point Group Classification (Accuracy %) | Computational Efficiency | Key Strengths |
|---|---|---|---|---|---|
| GCN | 0.68-0.72 [20] | Varies by dataset [5] | 85-89% [33] | Moderate [74] | Stable training, good baseline |
| GAT | 0.70-0.74 [20] | Varies by dataset [5] | 87-90% [33] | Lower due to attention [74] | Adaptive neighborhood weighting |
| GIN | 0.71-0.73 [20] | Competitive on small molecules [5] | 92.7% [33] | Moderate to High [74] | Maximum discriminative power for structures |
| MPNN | 0.75 (highest) [20] | Strong on quantum properties [5] | N/A | Varies by implementation [20] | Flexible framework, state-of-the-art on reaction yields |
Task-Specific Performance Analysis
Reaction Yield Prediction
In predicting yields for cross-coupling reactions (Suzuki, Sonogashira, Buchwald-Hartwig, etc.), MPNN achieves the highest predictive performance with an R² value of 0.75, outperforming other architectures. This superiority stems from MPNN's flexible message functions that can effectively model complex reaction mechanisms and transition metal catalysis [20]. The integrated gradients method applied to MPNN models has enhanced interpretability by identifying which molecular substructures most significantly impact predicted yields [20].
Molecular Property Prediction
For broad molecular property prediction benchmarks (including quantum chemical properties, solubility, and toxicity), architectural performance varies significantly by dataset. Recent innovations integrating Kolmogorov-Arnold Networks (KANs) into GNN frameworks have shown consistent improvements in both accuracy and computational efficiency. KA-GNN variants (KA-GCN and KA-GAT) replace standard MLP transformations with Fourier-based KAN modules in node embedding, message passing, and readout components, demonstrating enhanced function approximation capabilities [5].
Molecular Symmetry Classification
In predicting molecular point groups from 2D topological structures—critical for understanding spectroscopic properties and reactivity—GIN achieves the highest accuracy at 92.7% with an F1-score of 0.924 on the QM9 dataset. This superior performance directly results from GIN's theoretical foundation in graph isomorphism testing, enabling it to better capture both local connectivity and global structural information essential for symmetry determination [33].
Experimental Protocols and Methodologies
Standardized Benchmarking Framework
To ensure fair comparison across architectures, researchers should implement the following standardized experimental protocol:
Table 2: Key experimental components for comparative GNN evaluation
| Component | Specification | Purpose |
|---|---|---|
| Dataset | Multiple from MoleculeNet (QM9, FreeSolv, Tox21) [75] | Ensure diverse property coverage |
| Splitting | Scaffold split with 80:10:10 ratio | Evaluate generalization to novel structures |
| Node Features | Atomic number, degree, hybridization, valence, aromaticity [74] | Encode chemical identity |
| Edge Features | Bond type, conjugation, ring membership, spatial distance [74] | Encode bonding context |
| Optimization | Hyperparameter search (TPE, CMA-ES, Random Search) [75] | Ensure optimal configuration |
| Validation | Stratified k-fold cross-validation (k=5) | Robust performance estimation |
Implementation Details
For reproducible benchmarking, the following implementation specifications are recommended:
Graph Representation: Molecular graphs should include both covalent and, when available, non-covalent interactions, as the latter have been shown to significantly enhance prediction accuracy for certain properties [5].
Training Configuration: Use Adam optimizer with initial learning rate of 0.001 and early stopping based on validation loss with patience of 100 epochs. Batch size should be optimized for each architecture but typically ranges from 32-128.
Regularization: Apply L2 regularization (weight decay=1e-5) and dropout (rate=0.2-0.5) appropriate to model complexity, with higher rates for larger parameter models like GAT.
Message Passing Steps: Limit to 3-5 layers to avoid over-smoothing, with skip connections or residual blocks in deeper architectures.
The following diagram illustrates the complete experimental workflow from data preparation to model evaluation:
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential computational tools for GNN-based molecular property prediction
| Tool/Resource | Type | Function | Application Context |
|---|---|---|---|
| MoleculeNet | Benchmark Dataset Collection | Standardized molecular datasets with curated properties | Model evaluation and comparison [75] |
| CHEMBL | Chemical Database | Bioactivity data for drug discovery tasks | Few-shot learning for rare targets [50] |
| GRATIS Framework | Graph Representation Tool | Generates task-specific topology and multi-dimensional edge features | Handling non-graph data or enhancing existing graphs [76] |
| KA-GNN Implementation | Model Architecture | Integrates Kolmogorov-Arnold Networks with GNN components | Improved accuracy and interpretability [5] |
| Hyperparameter Optimization | Optimization Methods | TPE, CMA-ES algorithms for parameter tuning | Efficient model configuration [75] |
| Integrated Gradients | Interpretation Method | Attributes predictions to input features | Model explainability and chemical insight generation [20] |
Future Directions and Research Opportunities
The field of GNNs for molecular property prediction continues to evolve rapidly, with several promising research directions emerging:
Integration with Emerging Architectures
The successful integration of Kolmogorov-Arnold Networks (KANs) with GNN backbones demonstrates the potential of hybrid architectures. KA-GNNs replace standard MLPs with Fourier-based KAN modules in node embedding, message passing, and readout components, achieving superior accuracy and parameter efficiency while maintaining interpretability [5]. Future work could explore integration with other emerging architectural paradigms.
Few-Shot Learning for Molecular Property Prediction
Few-shot molecular property prediction (FSMPP) addresses the critical challenge of scarce experimental annotations, particularly for novel targets or rare diseases. Key research challenges include cross-property generalization under distribution shifts and cross-molecule generalization under structural heterogeneity [50]. Meta-learning approaches that leverage related properties and molecular structures show particular promise for real-world drug discovery applications where labeled data is limited.
Advanced Graph Representation Strategies
Traditional covalent-bond-based molecular graph representations have inherent limitations in capturing complex molecular interactions. Recent approaches incorporating non-covalent interactions and geometry-aware representations have demonstrated significant performance improvements [5]. The GRATIS framework, which generates task-specific topology and multi-dimensional edge features from any arbitrary input, represents another advancement that could be further specialized for molecular domains [76].
This comparative analysis demonstrates that GCN, GAT, GIN, and MPNN architectures each present distinct strengths and limitations for molecular property prediction tasks. MPNN achieves superior performance for reaction yield prediction, while GIN excels in symmetry-based classification tasks requiring structural discrimination. GAT's attention mechanism provides adaptive neighborhood weighting beneficial for heterogeneous molecular systems, and GCN remains a strong, computationally efficient baseline. The choice of architecture should be guided by specific task requirements, dataset characteristics, and interpretability needs. Future research directions including KA-GNN integration, few-shot learning approaches, and advanced graph representation strategies promise to further enhance the capabilities of GNNs in molecular property prediction, accelerating drug discovery and materials design.
Graph Neural Networks (GNNs) have emerged as powerful frameworks for learning from graph-structured data, achieving remarkable success across scientific domains. This case study examines the application of GNNs in two distinct yet challenging fields: predicting chemical reaction yields in organic chemistry and forecasting clinical outcomes in healthcare. By exploring these applications within the broader context of molecular property prediction research, we highlight both the transformative potential and practical implementation of GNN architectures. The ability of GNNs to natively operate on structured data—from molecular graphs to patient networks—makes them uniquely suited for these domains where relationships between entities are as crucial as the entities themselves.
The following sections provide a technical assessment of GNN performance across these domains, detailing experimental methodologies, quantitative results, and practical resources for researchers. We structure our analysis to enable direct comparison of approaches, architectures, and outcomes, with particular emphasis on recent advancements that push the boundaries of predictive accuracy and practical utility.
GNNs for Chemical Reaction Yield Prediction
Experimental Frameworks and Architectural Comparison
Research in chemical reaction yield prediction has evaluated multiple GNN architectures to identify optimal configurations for molecular graph processing. A comprehensive 2025 study assessed seven major GNN variants on diverse transition metal-catalyzed cross-coupling reactions including Suzuki, Sonogashira, Cadiot–Chodkiewicz, Ullmann-type, and Buchwald–Hartwig couplings [77].
The experimental protocol involved representing each reaction's molecular components as graphs, where atoms constitute nodes and bonds form edges. Node features encoded atom-specific properties (atom type, formal charge, degree, hybridization, valence, chirality, etc.), while edge features represented bond characteristics (bond type, stereochemistry, conjugation) [78]. The models were trained to map these graph representations to continuous yield values.
As shown in Table 1, Message Passing Neural Networks (MPNN) achieved superior performance, indicating their effectiveness at capturing complex molecular interactions crucial for yield prediction [77].
Table 1: Performance of GNN Architectures for Chemical Reaction Yield Prediction
| GNN Architecture | Performance (R²) | Key Characteristics |
|---|---|---|
| Message Passing Neural Network (MPNN) | 0.75 | Models iterative message exchange between nodes along edges [77] |
| Graph Isomorphism Network (GIN) | 0.71 | High expressive power for graph discrimination [77] |
| Graph Attention Network (GAT/GATv2) | 0.68-0.70 | Uses attention mechanisms to weight neighbor importance [77] |
| Residual Graph Convolutional Network (ResGCN) | 0.67 | Incorporates residual connections to train deeper networks [77] |
| Graph Sample and Aggregate (GraphSAGE) | 0.66 | Efficiently aggregates sampled neighbor information [77] |
| Graph Convolutional Network (GCN) | 0.65 | Basic spectral graph convolution operation [77] |
Advanced Architectures and Pre-training Strategies
Beyond standard architectures, researchers have developed specialized GNN frameworks to enhance molecular property prediction. Kolmogorov-Arnold GNNs (KA-GNNs) integrate Fourier-based learnable univariate functions into GNN components—node embedding, message passing, and readout operations—replacing traditional multilayer perceptrons (MLPs) [5]. This approach, grounded in the Kolmogorov-Arnold representation theorem, improves both prediction accuracy and computational efficiency while offering enhanced interpretability by highlighting chemically meaningful substructures [5].
To address data scarcity issues, novel pre-training strategies have emerged. MolDescPred defines a pre-text task where GNNs learn to predict molecular descriptors derived from large unlabeled molecular databases [78]. After applying principal component analysis (PCA) to reduce descriptor dimensionality, the model is pre-trained to predict the resulting principal component scores as pseudo-labels [78]. This approach significantly enhances performance on downstream yield prediction tasks, particularly when fine-tuning data is limited.
Another innovative framework integrates knowledge from Large Language Models (LLMs) with structural features from pre-trained molecular models [3]. By prompting LLMs like GPT-4o and DeepSeek-R1 to generate domain knowledge and executable code for molecular vectorization, researchers create knowledge-based features that complement structural representations, yielding state-of-the-art prediction performance [3].
Experimental Protocol for Yield Prediction
A standardized experimental methodology has emerged for GNN-based reaction yield prediction:
Data Representation: Represent reactants and products as molecular graphs where atoms are nodes (with feature vectors) and bonds are edges (with bond features) [78].
Model Selection: Implement and compare multiple GNN architectures (MPNN, GIN, GAT, GCN, etc.) using a consistent evaluation framework [77].
Interpretability Analysis: Apply explainable AI techniques like integrated gradients to determine contribution of input descriptors to yield predictions [77].
Evaluation: Use k-fold cross-validation and report R² values alongside other regression metrics on held-out test sets containing reactions not seen during training [77].
The following diagram illustrates the complete workflow for GNN-based chemical reaction yield prediction, integrating both standard and advanced approaches:
GNNs for Clinical Outcome Prediction
Clinical Application Domains and Performance
In healthcare, GNNs have demonstrated strong performance across multiple clinical prediction tasks by effectively modeling complex relationships in electronic health records (EHR) and patient networks. As surveyed in 2023, diagnosis prediction represents the most common application (72% of studies), with graph attention networks (GAT) emerging as the predominant architecture (38% of implementations) [79].
Clinical applications extend to specialized domains including specialty care recommendation, chronic disease prediction, and emergency department triage. In specialty care, GNN-based recommender systems achieved significant improvements over manual clinical checklists, with experimental results showing an 8% improvement in ROC-AUC for endocrinology (ROC-AUC=0.88) and 5% for hematology (ROC-AUC=0.84) [80]. For chronic disease prediction, GNNs with attention mechanisms reached 93.49% accuracy for cardiovascular disease and 89.15% for chronic pulmonary disease prediction [81].
Table 2: GNN Performance Across Clinical Prediction Tasks
| Clinical Domain | Prediction Task | Best Performance | Key GNN Architecture |
|---|---|---|---|
| Specialty Care | Procedure recommendation | ROC-AUC: 0.88 (Endo), 0.84 (Hemo) | Heterogeneous GNN [80] |
| Chronic Disease | Cardiovascular disease | Accuracy: 93.49% | GNN with attention [81] |
| Chronic Disease | Chronic pulmonary disease | Accuracy: 89.15% | GNN with attention [81] |
| Patient Outcome | Length of stay | Improved over LSTM baseline | LSTM-GNN hybrid [82] |
| Emergency Care | Triage prioritization | Outperformed traditional methods | Multiple GNN architectures [83] |
Methodologies for Clinical GNN Implementation
Clinical GNN implementation follows distinct methodological frameworks tailored to healthcare data structures:
Weighted Patient Network Framework: For chronic disease prediction, researchers construct weighted patient networks where patients form nodes connected by edges weighted according to clinical similarity [81]. The framework involves: (1) creating a patient-disease bipartite graph, (2) projecting to a patient-patient network with weights representing shared disease comorbidities, (3) applying GNNs to learn patient representations incorporating network structure, and (4) predicting disease risk using these enriched representations [81].
Patient Similarity Network for Triage: For emergency department triage, each patient is represented as a node with edges indicating similarity based on vital signs, symptoms, and medical history [83]. The graph is embedded into a latent space where a node classifier assigns triage priority levels, leveraging both patient attributes and relational information for more accurate prioritization than traditional methods [83].
Hybrid Temporal-Relational Models: For ICU outcome prediction, LSTM-GNN hybrids combine Long Short-Term Memory networks for processing physiological time series with GNNs that incorporate diagnostic relational information [82]. This approach connects similar patients in a graph structure, allowing the model to learn from neighborhood information and rarer disease patterns that might be overlooked in purely temporal models [82].
The following workflow diagram illustrates the generalized approach for GNN-based clinical outcome prediction:
Successful implementation of GNNs for molecular and clinical prediction requires specific data resources, software tools, and computational frameworks. Table 3 summarizes key resources mentioned in the research literature.
Table 3: Essential Research Resources for GNN Implementation
| Resource Category | Specific Resource | Description and Application |
|---|---|---|
| Chemical Data | Cross-coupling reaction datasets | Diverse datasets encompassing Suzuki, Sonogashira, and other coupling reactions with yield values [77] |
| Clinical Data | MIMIC-III | Publicly available critical care database commonly used for clinical prediction tasks [79] |
| Clinical Data | Institutional EHR data | De-identified electronic health records from healthcare institutions for specialty care prediction [80] |
| Molecular Tools | Mordred calculator | Calculates 1,826 molecular descriptors for pre-training GNNs [78] |
| Computational Framework | Graph Neural Network libraries | PyTor Geometric, DGL, or other GNN implementations supporting MPNN, GAT, GIN architectures [77] |
| Interpretability Tools | Integrated gradients | Method for determining contribution of input features to model predictions [77] |
| LLM Integration | GPT-4o, DeepSeek-R1 | Large language models for generating knowledge-based features to augment structural information [3] |
This technical assessment demonstrates that GNNs deliver strong performance across both chemical and clinical prediction domains, with architectural choices significantly impacting outcomes. In chemistry, Message Passing Neural Networks achieve superior yield prediction (R²=0.75), while graph attention networks dominate clinical applications. The integration of advanced techniques—including Fourier-based KA-GNNs, molecular descriptor pre-training, LLM knowledge fusion, and hybrid temporal-relational models—consistently enhances predictive accuracy and model interpretability.
Successful implementation requires careful attention to data representation, appropriate architectural selection, and domain-specific methodological adaptations. The experimental protocols and resources detailed herein provide researchers with practical guidance for developing GNN solutions across scientific domains, contributing to the broader thesis that graph representation learning offers powerful frameworks for molecular property prediction and beyond.
Conclusion
Graph Neural Networks have firmly established themselves as a powerful and versatile paradigm for molecular property prediction, fundamentally changing the landscape of computational drug discovery. By directly learning from molecular graph structures, GNNs like GCN, GAT, and the innovative KA-GNNs offer superior accuracy and interpretability over traditional descriptor-based methods. Successfully deploying these models requires carefully navigating challenges of data scarcity through few-shot learning and ensuring robust generalization. The future of GNNs in biomedicine is bright, pointing toward more expressive architectures, integration of 3D structural information, better few-shot and self-supervised learning techniques, and increased application in de novo drug design and clinical decision support systems. These advancements promise to further accelerate the identification of novel therapeutics and deepen our understanding of molecular mechanisms.
References
- 1. A compact review of molecular property prediction with ... [https://www.sciencedirect.com/science/article/pii/S1740674920300305]
- 2. Advancing Molecular Property Prediction for Environmental ... [https://www.sciencedirect.com/science/article/abs/pii/S0169743925002667]
- 3. Enhancing Molecular Property Prediction with Knowledge ... [https://arxiv.org/html/2509.20664v1]
- 4. Graph Property Prediction [https://snap-stanford.github.io/ogb-web/docs/graphprop/]
- 5. Kolmogorov–Arnold graph neural networks for molecular ... [https://www.nature.com/articles/s42256-025-01087-7]
- 6. Molecular property prediction in the ultra‐low data regime [https://www.nature.com/articles/s42004-025-01592-1]
- 7. A Graph Neural Network for Molecular Property Prediction ... [https://arxiv.org/abs/2410.12156]
- 8. FGBench: A Dataset and Benchmark for Molecular ... [https://arxiv.org/html/2508.01055v1]
- 9. Sharing new breakthroughs and artifacts supporting ... [https://ai.meta.com/blog/meta-fair-science-new-open-source-releases/]
- 10. Graph Neural Networks (GNNs) – Comprehensive Guide [https://viso.ai/deep-learning/graph-neural-networks/]
- 11. Graph Neural Networks (GNN) Explained [https://www.ultralytics.com/glossary/graph-neural-network-gnn]
- 12. Understanding Graph Neural Networks GNN in Depth [https://www.ai-futureschool.com/en/computing/understanding-graph-neural-networks-gnn-in-depth.php]
- 13. Understanding Message Passing in Graph Neural Networks [https://medium.com/@rajeev.chandran_61731/understanding-message-passing-frameworks-in-graph-neural-networks-944d9e2a1105]
- 14. What is a GNN? — ignnition main documentation [https://ignnition.org/doc/what_is_a_gnn.html]
- 15. Graph Neural Network: In a Nutshell | Karthick Panner Selvam [https://karthick.ai/blog/2024/Graph-Neural-Network/]
- 16. [2507.03430] Multi-Level Fusion Graph Neural Network for ... [https://arxiv.org/abs/2507.03430]
- 17. A Survey of Graph Neural Networks for Drug Discovery [https://arxiv.org/html/2509.07887v1]
- 18. A review of graph neural networks: concepts, architectures ... [https://journalofbigdata.springeropen.com/articles/10.1186/s40537-023-00876-4]
- 19. Geometry-enhanced molecular representation learning for ... [https://www.nature.com/articles/s42256-021-00438-4]
- 20. Performance assessment of various graph neural network ... [https://pubs.rsc.org/en/content/articlehtml/2025/cp/d5cp01072a]
- 21. Evaluating Machine Learning Models for Molecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12529777/]
- 22. Consistency-regularized graph neural networks for ... [https://pubmed.ncbi.nlm.nih.gov/41056596/]
- 23. Enhancing molecular property prediction with quantized GNN ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-00989-3]
- 24. Building A Graph Convolutional Network for Molecular ... [https://medium.com/data-science/building-a-graph-convolutional-network-for-molecular-property-prediction-978b0ae10ec4]
- 25. Message-passing neural network (MPNN) for molecular ... [https://keras.io/examples/graph/mpnn-molecular-graphs/]
- 26. T035 · GNN-based molecular property prediction [https://projects.volkamerlab.org/teachopencadd/talktorials/T035_graph_neural_networks.html]
- 27. Contrastive Dual-Interaction Graph Neural Network for ... [https://arxiv.org/html/2405.02628v1]
- 28. Chemistry-intuitive explanation of graph neural networks for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10160109/]
- 29. Quantitative evaluation of explainable graph neural ... [https://www.sciencedirect.com/science/article/pii/S2666389922002604]
- 30. Interpretability of Graph Neural Networks: An Exploratory ... [https://medium.com/the-quantastic-journal/interpretability-of-graph-neural-networks-an-exploratory-study-of-nodes-features-and-attention-70799aec74dd]
- 31. Multi-task methods for molecular property prediction [https://www.sciencedirect.com/science/article/pii/S0098135425002571]
- 32. Benchmarking OMol25-Trained Models on Experimental ... [https://rowansci.com/publications/omol25-redox]
- 33. A Method For Predicting Molecular Point Group Based On ... [https://www.sciencedirect.com/science/article/pii/S2949747725000144]
- 34. A simple and efficient graph Transformer architecture for ... [https://www.sciencedirect.com/science/article/abs/pii/S0009250923006139]
- 35. An end-to-end attention-based approach for learning on ... [https://www.nature.com/articles/s41467-025-60252-z]
- 36. MolGraphormer: An Interpretable GNN-Transformer for ... [https://chemrxiv.org/engage/chemrxiv/article-details/68f25b22bc2ac3a0e08f5f6d]
- 37. A self-conformation-aware pre-training framework for ... [https://www.nature.com/articles/s41467-025-59634-0]
- 38. GraphXForm: graph transformer for computer-aided molecular ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d4dd00339j]
- 39. KA-GNN: Kolmogorov-Arnold Graph Neural Networks for ... [https://arxiv.org/abs/2410.11323]
- 40. Applying graph neural network models to molecular ... [https://www.sciencedirect.com/science/article/pii/S2949747724000083]
- 41. Revolutionary Graph Neural Networks Predict Molecular ... [https://bioengineer.org/revolutionary-graph-neural-networks-predict-molecular-properties/]
- 42. Graph neural networks driven acceleration in drug discovery [https://www.sciencedirect.com/science/article/pii/S2211383525006884]
- 43. An enhanced deep learning framework for HIV drug ... [https://pubmed.ncbi.nlm.nih.gov/40617525/]
- 44. An enhanced deep learning framework for HIV drug ... [https://www.sciencedirect.com/science/article/abs/pii/S0024320525004709]
- 45. [2312.17506] A graph neural network-based model with ... [https://arxiv.org/abs/2312.17506]
- 46. Drug toxicity prediction model based on enhanced graph ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482524016998]
- 47. Graph Neural Network-Based Toxicity Prediction by ... [https://www.mdpi.com/2305-6304/13/11/953]
- 48. Equivariant Graph Neural Networks for Toxicity Prediction [https://pmc.ncbi.nlm.nih.gov/articles/PMC10583285/]
- 49. Efficient Learning of Molecular Properties Using Graph Neural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12631692/]
- 50. Few-shot Molecular Property Prediction: A Survey [https://arxiv.org/html/2510.08900v1]
- 51. Adaptive Transfer of Graph Neural Networks for Few-Shot ... [https://pubmed.ncbi.nlm.nih.gov/37878431/]
- 52. Few-shot Molecular Property Prediction: A Survey [https://arxiv.org/abs/2510.08900]
- 53. Attribute-guided prototype network for few-shot molecular ... [https://pubmed.ncbi.nlm.nih.gov/39133096/]
- 54. Knowledge-enhanced Relation Graph and Task Sampling ... [https://www.sciencedirect.com/science/article/abs/pii/S002002552500489X]
- 55. Using GNN property predictors as molecule generators [https://pmc.ncbi.nlm.nih.gov/articles/PMC12062463/]
- 56. Advancing Graph Neural Networks for Complex Relational ... [https://www.mdpi.com/2227-7390/13/18/2956]
- 57. A performance booster for Graph Neural Networks on node ... [https://www.sciencedirect.com/science/article/abs/pii/S0893608025001637]
- 58. Label Reusing Based Graph Neural Network for ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=4510873]
- 59. FragNet: A Graph Neural Network for Molecular Property ... [https://arxiv.org/html/2410.12156v1]
- 60. Hierarchical Molecular Graph Self-Supervised Learning for ... [https://www.nature.com/articles/s42004-023-00825-5]
- 61. Enhancing molecular property prediction through data ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-01103-3]
- 62. Predicting molecular properties based on the interpretable ... [https://pubmed.ncbi.nlm.nih.gov/36526280/]
- 63. MoleculeNet: a benchmark for molecular machine learning [https://pmc.ncbi.nlm.nih.gov/articles/PMC5868307/]
- 64. An Introduction To MoleculeNet [https://deepchem.io/tutorials/an-introduction-to-moleculenet/]
- 65. snap-stanford/ogb: Benchmark datasets, data loaders, and ... [https://github.com/snap-stanford/ogb]
- 66. Quantum chemistry structures and properties of 134 kilo ... [https://www.nature.com/articles/sdata201422]
- 67. torch_geometric.datasets.QM9 - PyTorch Geometric [https://pytorch-geometric.readthedocs.io/en/2.6.1/generated/torch_geometric.datasets.QM9.html]
- 68. The Complete Beginner's Guide to Graph Neural Networks ... [https://ikala.ai/blog/ai-trends/the-complete-beginners-guide-to-graph-neural-networks-gnns/]
- 69. Metrics in Machine Learning and Computer Vision | ASR Engineering [https://adilsarsenov.dev/posts/metrics/]
- 70. Could graph neural networks learn better molecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7888189/]
- 71. Performance metrics in Classification and Regression [https://iq.opengenus.org/performance-metrics-in-classification-regression/]
- 72. Graph neural networks with molecular segmentation for ... [https://www.sciencedirect.com/science/article/abs/pii/S0098135423002739]
- 73. Graph GNNs, 2025: Sparse Tricks That Matter [https://medium.com/@ThinkingLoop/graph-gnns-2025-sparse-tricks-that-matter-ffce33ce503c]
- 74. Benchmarking Graph Representations and Graph Neural ... [https://arxiv.org/html/2501.08305v2]
- 75. A Systematic Comparison Study on Hyperparameter ... [https://arxiv.org/abs/2102.04283]
- 76. GRATIS: Deep Learning Graph Representation with Task ... [https://arxiv.org/abs/2211.12482]
- 77. Performance assessment of various graph neural network ... [https://pubs.rsc.org/en/content/articlelanding/2025/cp/d5cp01072a]
- 78. Improving chemical reaction yield prediction using pre-trained ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00818-z]
- 79. Graph neural networks for clinical risk prediction based on ... [https://www.sciencedirect.com/science/article/pii/S1532046424000340]
- 80. Graph-Based Clinical Recommender: Predicting Specialists ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12140122/]
- 81. A weighted patient network-based framework for predicting ... [https://www.nature.com/articles/s41598-021-01964-2]
- 82. Predicting Patient Outcomes with Graph Representation ... [https://arxiv.org/abs/2101.03940]
- 83. Leveraging graph neural networks for supporting automatic ... [https://www.nature.com/articles/s41598-024-63376-2]