Research Articles
Validation Metrics for Continuous Variables: A 2025 Guide for Robust Biomedical Research and Drug Development
This article provides a comprehensive framework for selecting, applying, and interpreting validation metrics for continuous variables in biomedical and clinical research.
Statistical Tests for Model Accuracy Assessment: A Comprehensive Guide for Biomedical Research
This article provides a comprehensive framework for assessing the statistical accuracy of predictive models in biomedical and clinical research.
Cross-Validation in Computational Science: A Comprehensive Guide for Biomedical Researchers
This article provides a comprehensive guide to cross-validation techniques tailored for researchers, scientists, and professionals in drug development and computational science.
Bridging the Digital and Physical: A Strategic Framework for Comparing Model Predictions with Experimental Data in Biomedical Research
This article provides a comprehensive guide for researchers and drug development professionals on the critical process of validating computational models against experimental data.
AIC vs BIC: A Researcher's Guide to Optimal Model Selection in Drug Development
This article provides a comprehensive guide to Akaike (AIC) and Bayesian (BIC) Information Criterions for researchers and professionals in drug development and biomedical sciences.
Goodness-of-Fit Tests for Computational Models: A Comprehensive Guide for Biomedical Research
This article provides a comprehensive guide to goodness-of-fit (GOF) tests for computational models, tailored for researchers, scientists, and professionals in drug development.
Navigating the Unknown: Advanced Computational Strategies for Deep Uncertainty in Drug Development
This article provides a comprehensive guide to computational strategies for decision-making under deep uncertainty (DMDU), tailored for researchers and professionals in drug development.
Bayes Factor Model Comparison: A Computational Guide for Biomedical Research and Drug Development
This article provides a comprehensive guide to Bayes Factor model comparison for researchers, scientists, and professionals in computational fields and drug development.
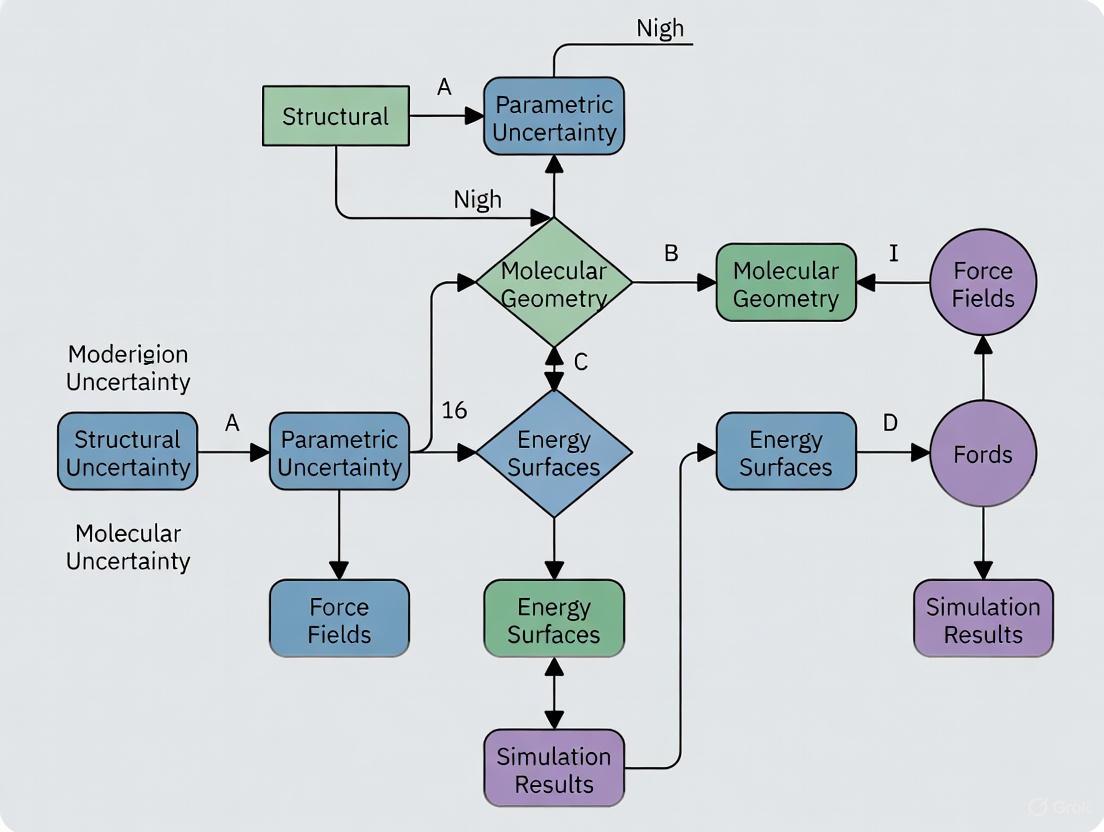
Structural vs. Parametric Uncertainty: A Framework for Robust Models in Drug Development
This article provides a comprehensive guide for researchers and drug development professionals on distinguishing and managing structural and parametric uncertainties in biomedical models.
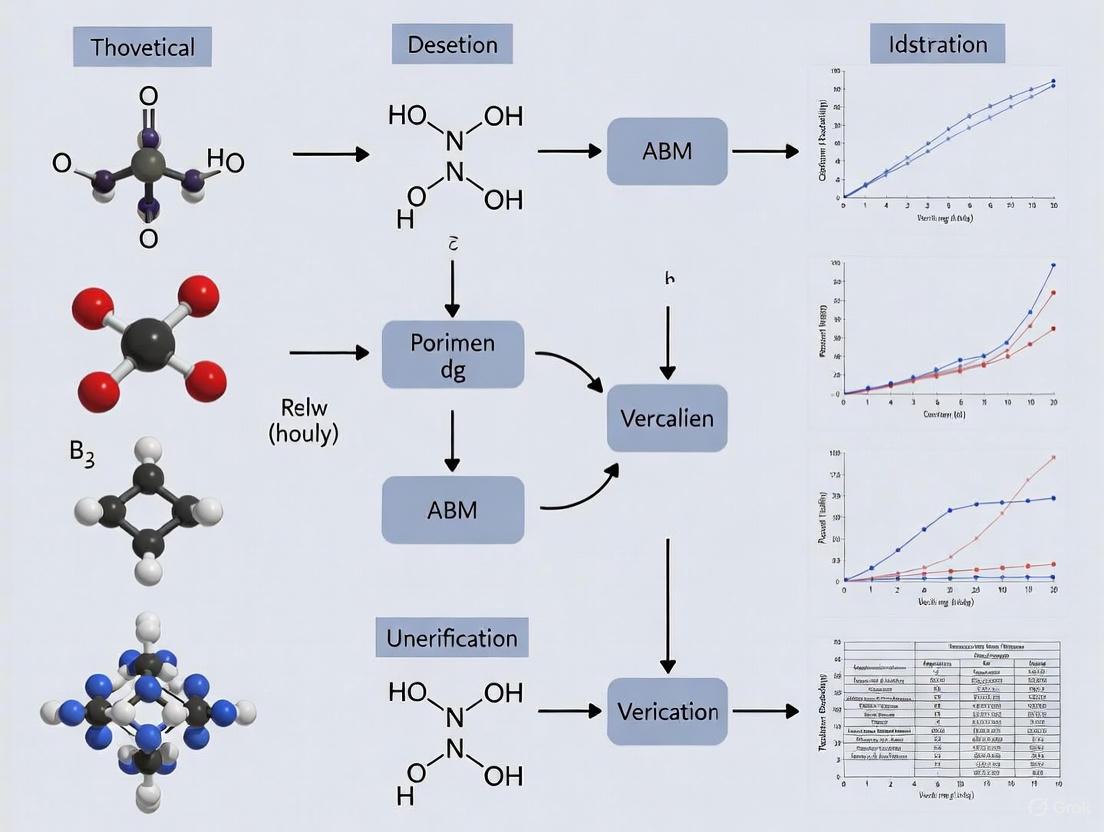
Existence and Uniqueness Analysis in ABM Verification: A Foundational Framework for Credible Computational Models in Biomedicine
This article provides a comprehensive guide to existence and uniqueness analysis, a critical but often overlooked component of Agent-Based Model (ABM) verification.